Introduction: A Protocol Written Before the AI Era
The Cartagena Protocol on Biosafety, adopted in January 2000 and entering into force in September 2003, was crafted in a world where the dominant biosafety concern was the cross-border movement of genetically modified crops. Its 40 articles and three annexes were designed to regulate the advance informed agreement procedure for living modified organisms (LMOs), the risk assessment framework under Annex III, and the Biosafety Clearing-House (BCH) information-sharing architecture. What the Protocol's architects could not have anticipated was a future in which artificial intelligence would simulate 500 million years of protein evolution in a single model run, design novel organisms from digital sequence information alone, and compress the design-build-test-learn cycle from years to days.
That future is now. The twelfth meeting of the Conference of the Parties serving as the meeting of the Parties to the Cartagena Protocol on Biosafety (COP-MOP 12) is scheduled for 19–30 October 2026 in Yerevan, Armenia. It arrives at a moment when the Protocol's foundational assumptions — that LMOs are produced through identifiable physical processes, that risk assessment can be conducted case-by-case on the basis of known donor organisms, and that the BCH can track movements through paper-based notifications — are being systematically undermined by AI-enabled synthetic biology. This post sets out the case for a structured reform agenda and proposes seven concrete interventions that Parties could adopt at Yerevan.
The AI-Synthetic Biology Convergence: What Has Changed Since 2003
The convergence of large language models (LLMs), autonomous biofoundries, and digital sequence information (DSI) has produced a qualitative shift in the biosafety landscape that the Protocol was not designed to address.
AI-driven protein and pathway design has moved from theoretical possibility to operational reality. The ESM3 language model, described in Science in 2024, generated a fluorescent protein with only 58% sequence identity to any known natural protein — a result that would have required decades of directed evolution using conventional methods. Autonomous enzyme engineering platforms, reported in Nature Communications in 2025, now execute design-build-test-learn cycles without human intervention at each step. The OECD's 2025 forward-looking analysis on synthetic biology and AI automation identifies this convergence as the single most consequential development for biosafety governance since the Protocol's adoption.
Digital sequence information has dissolved the physical boundary that the Protocol's advance informed agreement procedure was built around. Under the Protocol's current framework, the trigger for regulatory oversight is the transboundary movement of a physical LMO. But when an AI model trained on global genetic sequence databases designs a novel organism entirely in silico — and that design is then synthesised locally in a destination country — no physical LMO crosses a border. The CBD's 2024 SBSTTA-26 document on DSI explicitly acknowledges this governance gap, noting that "challenges may arise from the facilitated use of digital sequence information of genetic resources through the wide availability of AI tools." The Kunming-Montreal Global Biodiversity Framework's Target 13, which addresses fair and equitable benefit-sharing from DSI, does not resolve the biosafety dimension of this problem.
New genomic techniques (NGTs) — including base editing, prime editing, and epigenome editing — produce organisms that may be phenotypically indistinguishable from conventionally bred varieties yet carry targeted molecular changes. The EU's proposed NGT regulation, assessed against the Protocol by Vöneky et al. (2025) and critiqued by Testbiotech (2025), illustrates the tension: the EU Council proposal would allow environmental release of NGT Category 1 plants without mandatory risk assessment, a position that Testbiotech argues does not fulfil the Protocol's Annex III obligations.
AI-enabled biosecurity risks have been extensively documented in the 2026 CBD Open-Ended Online Forum on Synthetic Biology. Participants from the African Society for Synthetic Biology, UNEP, and multiple national focal points have highlighted that AI systems capable of simulating evolutionary processes can, in principle, be used to design enhanced pathogens or novel toxins — the dual-use concern that biosafety and biosecurity governance frameworks have historically treated as separate domains.
| Governance Gap | Protocol Article Affected | AI Driver |
|---|---|---|
| DSI-only organism design bypasses AIA procedure | Articles 7–10 (AIA) | LLM-based protein/pathway design |
| Risk assessment framework does not address AI-designed novelty | Annex III | Autonomous DBTL platforms |
| BCH notification system is paper-based and retrospective | Article 20 (BCH) | Rapid synthesis from DSI |
| No dual-use/biosecurity provisions | None | AI-accelerated pathogen design |
| Capacity asymmetry: African states cannot assess AI-designed LMOs | Article 22 (Capacity Building) | Biofoundry automation |
| NGT classification ambiguity | Article 3 (Definitions) | CRISPR, base editing, prime editing |
| Benefit-sharing gap for AI-derived DSI | Nagoya Protocol interface | AI training on biodiversity databases |
The COP-MOP 11 Baseline: What Yerevan Inherits
COP-MOP 11, held in Cali, Colombia in October 2024, produced a decisions booklet (published in all six UN official languages in February 2026) that provides the baseline from which Yerevan must advance. Key decisions from Cali relevant to the AI reform agenda include the continuation of the AHTEG on synthetic biology, the peer review process for the independent scientific study on synthetic biology (notified by the Secretariat in March 2026), and the ongoing work programme on risk assessment of LMOs containing engineered gene drives.
The CBD Secretariat's notification of 14 April 2026 (2026-038) calls for submissions on experiences with the additional voluntary guidance materials for case-by-case risk assessment of gene drive organisms — a process that will feed directly into COP-MOP 12 deliberations. The CBD's Open-Ended Online Forum on Synthetic Biology (February–March 2026, Thread 3492) has already surfaced the key technical concerns that Parties will need to address.
The African Dimension: Structural Disadvantage in the AI Era
Africa's position in the Cartagena Protocol governance architecture is structurally disadvantaged in ways that AI-synthetic biology convergence will intensify rather than ameliorate. The continent hosts 44 of the Protocol's 173 Parties, yet African national biosafety authorities collectively lack the technical infrastructure to assess AI-designed LMOs.
The African Society for Synthetic Biology's contribution to the 2026 CBD Online Forum explicitly identifies the AI-driven design of biological molecules from global genetic sequence databases as a threat to Africa's benefit-sharing rights under Target 13 of the KMGBF — noting that companies may design valuable products using biodiversity-derived sequence data from African ecosystems without equitable compensation to source countries or indigenous communities.
The Addis Ababa Declaration on Biosafety, adopted by African civil society organisations and scientists in 2024, called for a moratorium on AI-designed LMO releases in Africa pending the development of continent-specific risk assessment frameworks. The Africa CDC's 2025 health security framework, which integrates AI surveillance for biological threats, provides a model for what Protocol-level capacity building in the AI era could look like.
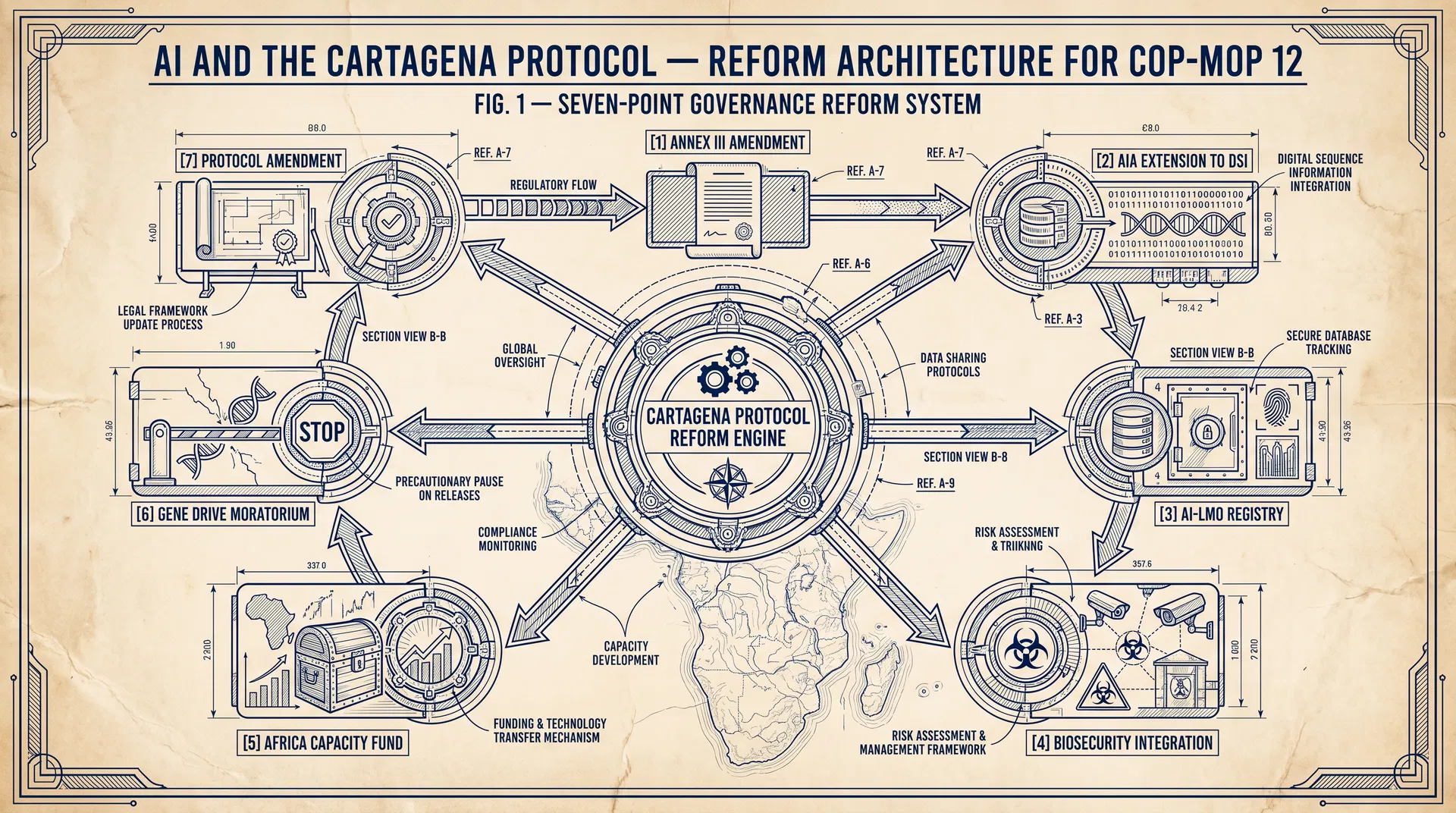
A Seven-Point Reform Agenda for COP-MOP 12
1. Amend Annex III to Address AI-Designed Organisms. COP-MOP 12 should mandate the AHTEG to develop supplementary guidance for AI-designed LMOs that addresses: (a) the absence of a conventional donor-recipient relationship; (b) the use of AI-predicted rather than empirically measured phenotypic data; and (c) the assessment of emergent properties not present in any natural organism.
2. Extend the AIA Procedure to Cover DSI-Triggered Synthesis. COP-MOP 12 should adopt a decision clarifying that the AIA procedure applies when an AI-designed LMO is synthesised in a destination country using DSI that originated from a source country's genetic resources — regardless of whether any physical LMO crosses a border.
3. Establish a Real-Time AI-LMO Registry in the BCH. COP-MOP 12 should mandate the development of a real-time registry of AI-designed LMOs that captures: design provenance; synthesis location; intended use; and risk assessment status. The registry should be interoperable with the Nagoya Protocol's ABS Clearing-House.
4. Integrate Biosecurity into the Protocol's Risk Assessment Framework. COP-MOP 12 should adopt a decision requesting the AHTEG to develop guidance on dual-use risk assessment for AI-designed LMOs, drawing on the Biological Weapons Convention's technical secretariat expertise.
5. Establish a Protocol-Level AI Capacity-Building Fund for African Parties. Article 22 capacity-building obligations should be operationalised through a dedicated AI biosafety capacity fund, capitalised at a minimum of USD 50 million over five years, targeting African national biosafety authorities.
6. Adopt a Precautionary Moratorium on Environmental Release of AI-Designed Gene Drive Organisms. COP-MOP 12 should adopt a decision extending the existing voluntary guidance on gene drive risk assessment to include a precautionary moratorium on environmental release of AI-designed gene drive organisms pending the development of the supplementary Annex III guidance.
7. Negotiate a Protocol Amendment to Define "AI-Designed Living Modified Organism." COP-MOP 12 should initiate a formal amendment process to add a definition of "AI-designed LMO" that captures organisms whose genetic sequences were generated by AI systems, regardless of the physical modification technique subsequently used to produce them.
The Road to Yerevan: Process and Politics
The political feasibility of this reform agenda depends on three factors: the AHTEG scientific study findings, the African Group's negotiating position, and the EU-Africa regulatory alignment question on NGTs. A COP-MOP 12 decision that explicitly reaffirms the Protocol's applicability to NGT organisms — including those produced using AI-assisted CRISPR design — would resolve this ambiguity in favour of the Protocol's precautionary mandate.
Conclusion: The Cartagena Protocol at a Crossroads
The Cartagena Protocol on Biosafety has survived 23 years as the primary international instrument for LMO governance because its core precautionary logic remains sound. What has changed is not the logic but the technology: AI has made it possible to design organisms of unprecedented novelty at unprecedented speed, using digital information that crosses no physical borders and triggers no existing regulatory procedure.
COP-MOP 12 in Yerevan is the moment for reform. For African Parties in particular, the stakes are existential — the continent's biodiversity is among the most valuable on Earth, and without a reformed Protocol that closes the DSI governance gap, extends the AIA procedure to AI-designed organisms, and builds African capacity to assess them, that biodiversity will be exploited without consent, without compensation, and without the precautionary safeguards that the Protocol was designed to provide.
The reform agenda begins at Yerevan. It cannot wait for COP-MOP 13.
Frequently Asked Questions
What is the Cartagena Protocol on Biosafety? The Cartagena Protocol on Biosafety is an international treaty under the Convention on Biological Diversity that regulates the transboundary movement of living modified organisms (LMOs). Adopted in 2000 and in force since 2003, it requires exporting countries to obtain advance informed agreement from importing countries before shipping LMOs, and mandates risk assessments under Annex III before LMOs are released into the environment.
What is COP-MOP 12 and when does it take place? COP-MOP 12 is the twelfth meeting of the Conference of the Parties serving as the meeting of the Parties to the Cartagena Protocol on Biosafety. It is scheduled for 19–30 October 2026 in Yerevan, Armenia.
Why does AI create a governance gap in the Cartagena Protocol? The Protocol's advance informed agreement procedure is triggered by the transboundary movement of a physical LMO. When an AI model designs an organism entirely from digital sequence information and that design is synthesised locally, no physical LMO crosses a border — so the AIA procedure is never triggered.
What is digital sequence information (DSI) and why does it matter for biosafety? Digital sequence information refers to genetic sequence data stored in databases such as GenBank and BOLD. AI models trained on DSI can design novel organisms without accessing physical genetic material from source countries, creating both a benefit-sharing gap and a biosafety governance gap.
What does the reform agenda mean for Africa? Africa hosts 44 of the Protocol's 173 Parties and contains some of the world's most biodiverse ecosystems. The reform agenda proposes a dedicated AI capacity-building fund and an extension of the AIA procedure to DSI-triggered synthesis to address Africa's structural disadvantage in the AI era of synthetic biology governance.
References
- Keiper, F. & Atanassova, A. (2025). International synthetic biology policy developments and implications for global biodiversity goals. Frontiers in Synthetic Biology, 3. https://doi.org/10.3389/fsybi.2025.1585337
- Testbiotech (2025). Comment on the report "Compatibility of the EU proposal for a regulation on plants based on certain new genomic techniques with the Cartagena Protocol on Biosafety." Background 30-4-2025.
- CBD Secretariat (2026). Open-Ended Online Forum on Synthetic Biology 2026, Topic 2: Most recent technological developments. Thread 3492.
- CBD Secretariat (2024). SBSTTA-26/4: Digital sequence information on genetic resources.
- CBD Secretariat (2026). Notification 2026-038: Submissions on experiences with voluntary guidance for risk assessment of gene drive organisms.