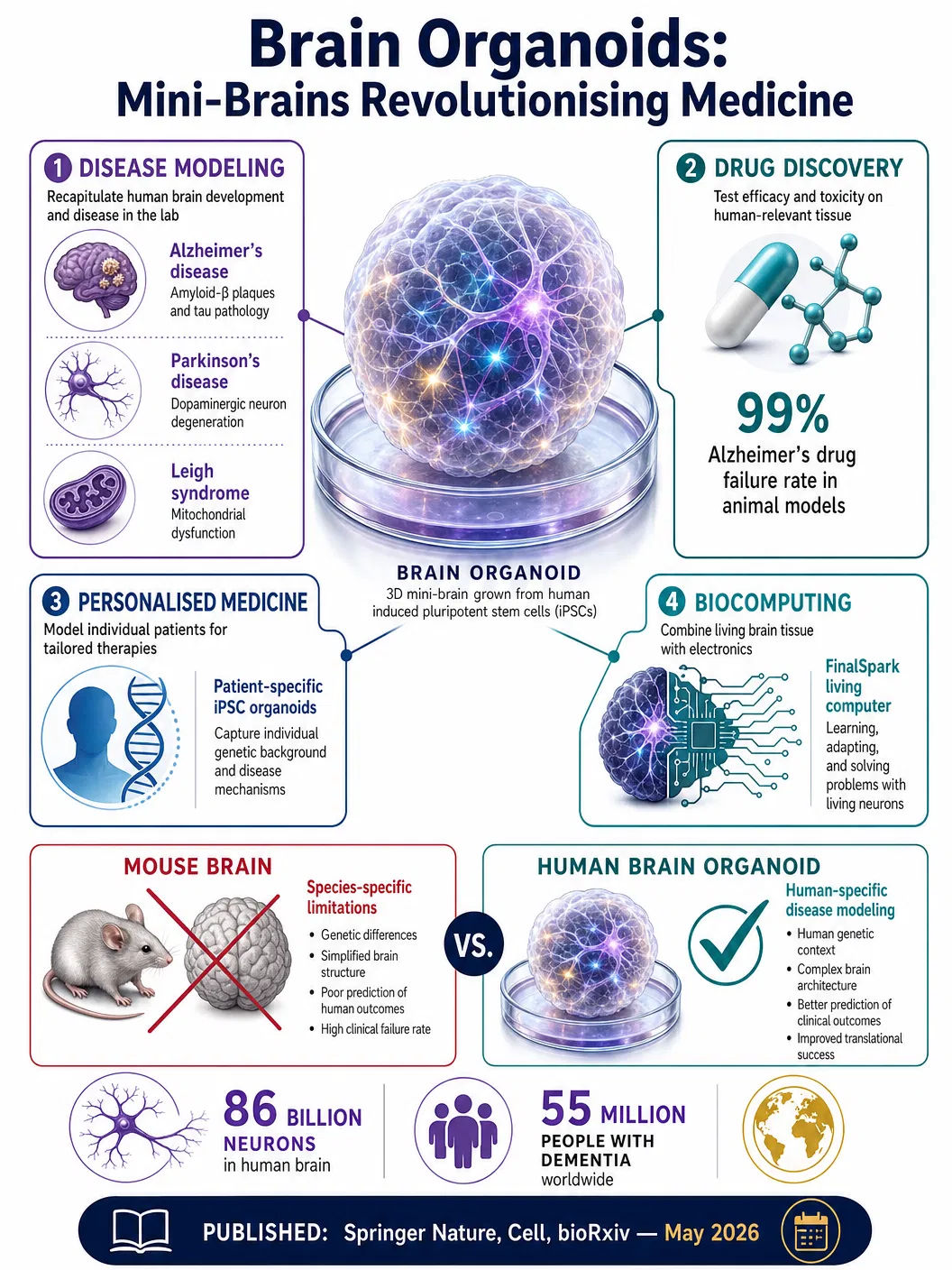
The human brain remains the most complex structure in the known universe — approximately 86 billion neurons connected by trillions of synapses, organised into circuits that produce consciousness, memory, emotion, and thought. Understanding how this organ develops, functions, and fails in disease has been one of the defining challenges of modern science. Animal models, particularly mice, have provided invaluable insights, but they carry a fundamental limitation: the mouse brain is not the human brain. Drugs that cure Alzheimer’s disease in mice have failed repeatedly in human clinical trials, contributing to a ninety-nine percent failure rate for Alzheimer’s therapeutics over the past two decades.
In May 2026, a convergence of publications in Springer Nature, Cell Stem Cell Reports, and multiple preprint servers has brought renewed attention to a technology that is fundamentally changing this equation: brain organoids. These three-dimensional structures, grown from human induced pluripotent stem cells (iPSCs) in laboratory dishes, self-organise into miniature approximations of brain tissue — complete with neurons, astrocytes, oligodendrocytes, and, in the most advanced versions, microglia-like immune cells. They are not brains. They do not think or feel. But they recapitulate key aspects of human brain development and disease in ways that no animal model can match.
A comprehensive review published on 12 May 2026 in Springer Nature’s Stem Cell Reviews and Reports examined the current state of iPSC-derived brain organoids as platforms for studying viral and toxicant-associated neurodegeneration. The review documented how organoids have been used to model the neurological effects of Zika virus, SARS-CoV-2, herpes simplex virus, and environmental neurotoxins — conditions that are difficult or impossible to study in animal models because the pathogens and toxins interact differently with human and animal neural tissue.
The drug discovery applications are particularly compelling. Traditional pharmaceutical development for neurological diseases follows a linear pipeline: identify a target, screen compounds in cell cultures, test promising candidates in animal models, and then advance to human clinical trials. The problem is that the animal model stage introduces a species-specific filter that eliminates many compounds that would work in humans while advancing others that ultimately fail. Brain organoids offer a human-specific screening platform that can be inserted earlier in the pipeline, potentially reducing the astronomical failure rates that have plagued neurological drug development.
A study published in PubMed Central in May 2026 demonstrated this principle for Leigh syndrome, a devastating mitochondrial disease that affects children and has no approved treatment. Researchers used brain organoids derived from patients’ own iPSCs to screen a library of repurposable drugs, identifying compounds that rescued mitochondrial function in the patient-specific organoids. This approach — using a patient’s own cells to test drugs before administering them — represents the ultimate expression of personalised medicine.
The technology is advancing rapidly on multiple fronts. Neuroimmune assembloids, described in a bioRxiv preprint from April 2026, integrate iPSC-derived hematopoietic progenitor cells into developing brain organoids, enabling them to mature into microglia-like immune cells that persist within the tissue. This innovation allows researchers to study neuroinflammation — a process implicated in Alzheimer’s, Parkinson’s, multiple sclerosis, and traumatic brain injury — in a fully human context for the first time.
Perhaps most provocatively, brain organoids are being explored as biological computing substrates. FinalSpark, a Swedish research group, has created what they describe as a “living computer” built from human brain organoids — tiny clusters of lab-grown neural tissue that can process information and respond to electrical stimulation. While true biological computing remains in its infancy, the concept suggests that brain organoids may have applications far beyond medicine, potentially contributing to the development of neuromorphic computing architectures that are more energy-efficient than silicon-based systems.
The commercial landscape is expanding accordingly. A PatSnap technology landscape report from April 2026 documented a surge in patent filings related to brain organoid technology, spanning disease modelling, drug screening, neuro-oncology, and biocomputing. Major pharmaceutical companies including Roche, Novartis, and AstraZeneca have established organoid programmes, and a growing ecosystem of biotech startups is developing organoid-based platforms for contract research and clinical applications.
Challenges remain. Current brain organoids lack the vascular networks that supply oxygen and nutrients to real brain tissue, limiting their size and maturation. They do not replicate the full complexity of brain architecture, including the layered cortical structure and long-range connectivity that characterise the adult human brain. And standardisation remains an issue: organoids grown in different laboratories using different protocols can vary significantly in their cellular composition and functional properties.
Despite these limitations, the trajectory is clear. Brain organoids are not replacing animal models overnight, but they are increasingly being recognised as an essential complement — and in some cases, a superior alternative — for studying human neurological disease and discovering new therapeutics. For the estimated 55 million people worldwide living with dementia, and the millions more affected by Parkinson’s, ALS, and rare neurological conditions, this technology represents a fundamentally new approach to understanding and treating the diseases that rob them of their minds.
References: iPSC-Derived 3D Brain Organoids as Next-generation Platforms to Study Viral and Toxicant-associated Neurodegeneration. Springer Nature Stem Cell Reviews and Reports. 12 May 2026. Menacho C, et al. Accelerating Leigh syndrome drug discovery through deep phenotyping of brain organoids. PubMed Central. 2026. Ruiz-Formoso I, et al. An Integrated Neuroimmune Assembloid Model. bioRxiv. 28 April 2026. PatSnap. Brain Organoid Technology Landscape 2026. April 2026. Cell Stem Cell Reports. Human organoids: Fit for drug discovery? 2026.