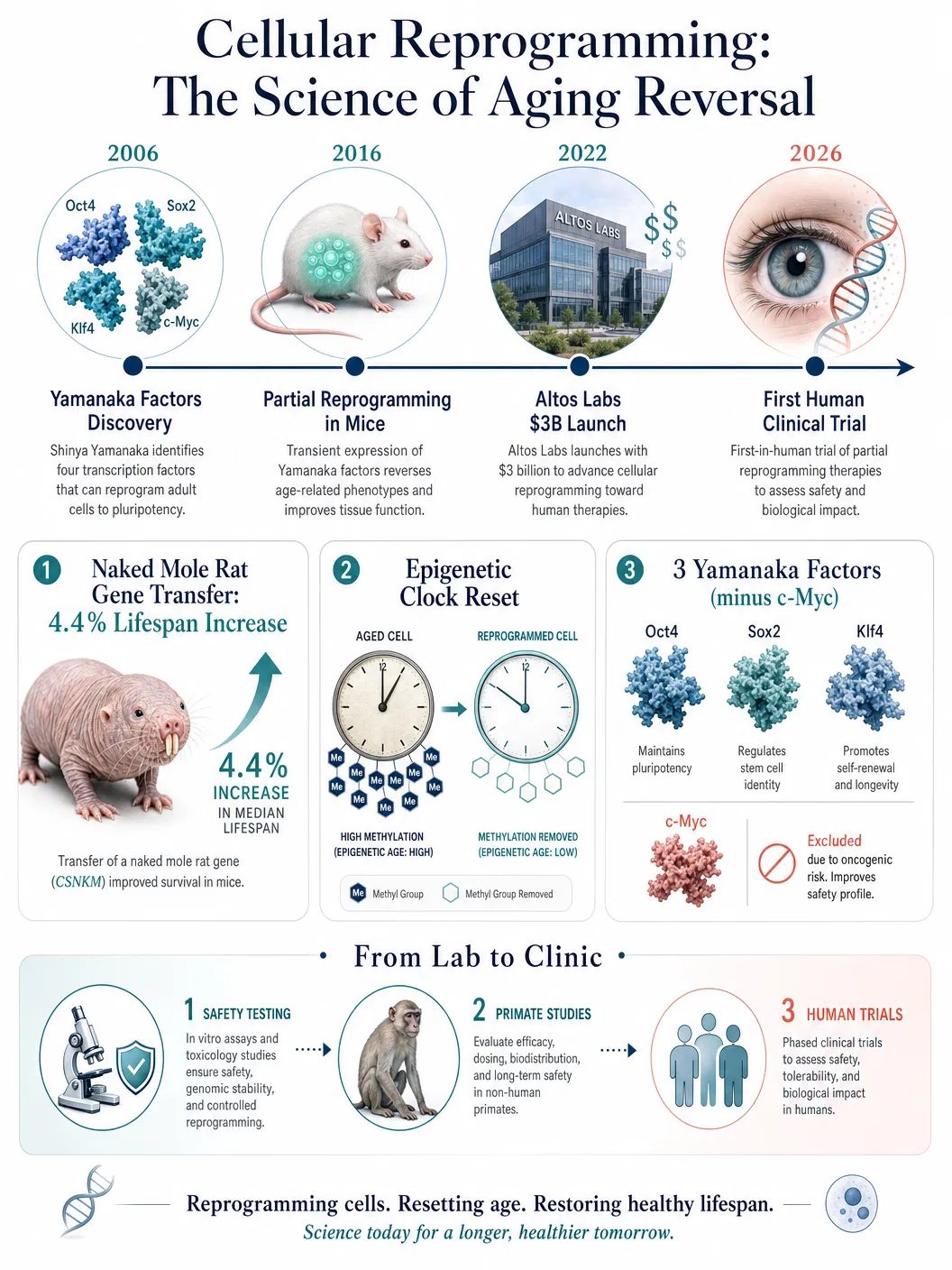
For most of human history, aging has been understood as an immutable biological process — a one-way journey from youth to senescence that no intervention could reverse. That understanding is now being challenged at the most fundamental level of biology. In 2026, Life Biosciences is preparing to launch the first human clinical trial of partial cellular reprogramming, a technique that uses a subset of Yamanaka transcription factors to dial back the developmental clock of aged cells without erasing their identity. If successful, this trial will not merely treat a single disease; it will validate the principle that biological aging itself is a modifiable condition.
The intellectual foundation for this moment was laid two decades ago when Shinya Yamanaka and his colleague at Kyoto University demonstrated that four transcription factors — Oct4, Sox2, Klf4, and c-Myc, collectively known as the Yamanaka factors — could reprogram adult cells into induced pluripotent stem cells capable of adopting entirely new identities. That discovery, which earned Yamanaka the Nobel Prize in 2012, opened the door to regenerative medicine. But a more radical implication lurked within the finding: if cells could be fully reprogrammed to an embryonic state, could they also be partially reprogrammed to a younger version of their current identity?
The answer, emerging from a decade of increasingly sophisticated mouse studies, appears to be yes. In 2016, Juan Carlos Izpisúa Belmonte and colleagues at the Salk Institute demonstrated that cyclic expression of Yamanaka factors extended the lifespan of mice with progeria, a condition of accelerated aging, and improved tissue regeneration in normal aged mice. Subsequent studies showed that partial reprogramming could rejuvenate skin cells, boost muscle regeneration, improve cardiac repair after injury, and even enhance memory performance in aged mice when applied to engram cells — the neurons responsible for encoding and storing memories. A study published in February 2026 demonstrated that partially reprogramming engram cells in mice with an Alzheimer's-like condition not only improved memory but reset the aberrant epigenetic signatures that had accumulated during the disease to a more normal state.
The clinical trial being prepared by Life Biosciences builds on work by Yuancheng Ryan Lu and David Sinclair at Harvard Medical School. Their approach uses a viral vector to deliver three of the four Yamanaka factors — excluding c-Myc, which at high levels can promote cancer — into the retinal nerve cells of patients with glaucoma. The genes are regulated by a genetic switch activated only when participants take a specific antibiotic, providing a safety mechanism to control the duration and intensity of reprogramming. Studies in non-human primates have shown no evidence of tumour formation or other adverse effects, and participants will be monitored for at least five years. The trial will initially enrol up to twelve patients with a specific type of glaucoma and up to six patients with non-arteritic anterior ischaemic optic neuropathy, a condition causing acute optic nerve damage.
The epigenetic dimension of aging provides the theoretical framework for why partial reprogramming works. Throughout life, the epigenome — the collection of chemical modifications, particularly methyl groups, that sit atop the genome and regulate gene activity — undergoes systematic shifts. These epigenetic changes are not random noise; they follow predictable patterns that constitute what researchers now call the "epigenetic clock." Partial reprogramming appears to reset portions of this clock, restoring gene expression patterns characteristic of younger cells without erasing the cell's fundamental identity. Lu, Sinclair, and their colleagues found that the rejuvenation of retinal nerve cells required two specific enzymes that remove methyl groups from DNA, directly linking the therapeutic effect to epigenetic remodelling.
Meanwhile, a parallel line of research is drawing inspiration from nature's own longevity champions. In May 2026, researchers at the University of Rochester reported the successful transfer of a longevity-related gene from the naked mole rat — an animal that can live over thirty years, roughly ten times longer than similarly sized rodents — into mice. The gene enhances production of high molecular weight hyaluronic acid, a substance that appears to protect against cancer, reduce chronic inflammation, and support healthier aging. The genetically modified mice showed stronger resistance to tumours, healthier gut function, and lower levels of age-related inflammation, with an approximate 4.4 percent increase in median lifespan. While modest in absolute terms, the finding demonstrates that longevity mechanisms evolved in one species can be functionally transferred to another — a principle with profound implications for human medicine.
The commercial landscape surrounding cellular reprogramming reflects the scale of scientific ambition. Altos Labs launched in 2022 with three billion dollars in funding from internet entrepreneur Yuri Milner and other investors, setting a world record for biotechnology startup financing. Sam Altman, the chief executive of OpenAI, invested in Retro Biosciences, which is pursuing partial reprogramming alongside other longevity approaches. Brian Armstrong, the chief executive of Coinbase, co-founded NewLimit, another reprogramming-focused company. The convergence of Silicon Valley capital and aging biology has created an ecosystem of unprecedented scale, though it has also raised concerns about hype outpacing evidence.
The risks of partial reprogramming are as significant as its promises. Push a cell too far toward its embryonic state and it may lose its functional identity entirely, potentially becoming cancerous. The removal of c-Myc from the reprogramming cocktail addresses one known oncogenic risk, but the long-term consequences of leaving partially reprogrammed cells in the human body remain unknown. As Tamir Chandra of the Mayo Clinic has cautioned, when cells lose their identity, danger follows. The upcoming clinical trial is designed primarily to assess safety, not efficacy — a necessary first step that reflects the scientific community's awareness that the gap between rejuvenating mouse retinal cells and safely treating human patients remains substantial.
What makes this moment in longevity science genuinely unprecedented is not any single experiment but the convergence of multiple independent lines of evidence — from Yamanaka factor reprogramming to cross-species gene transfer to epigenetic clock research — all pointing toward the same conclusion: biological aging is not a fixed programme but a malleable process that can, in principle, be slowed, halted, or partially reversed. Whether this principle translates into safe, effective human therapies will depend on the results of trials that are only now beginning. The science of aging has entered its clinical era, and the next few years will determine whether the promise of cellular rejuvenation becomes medical reality or remains a tantalising biological curiosity.