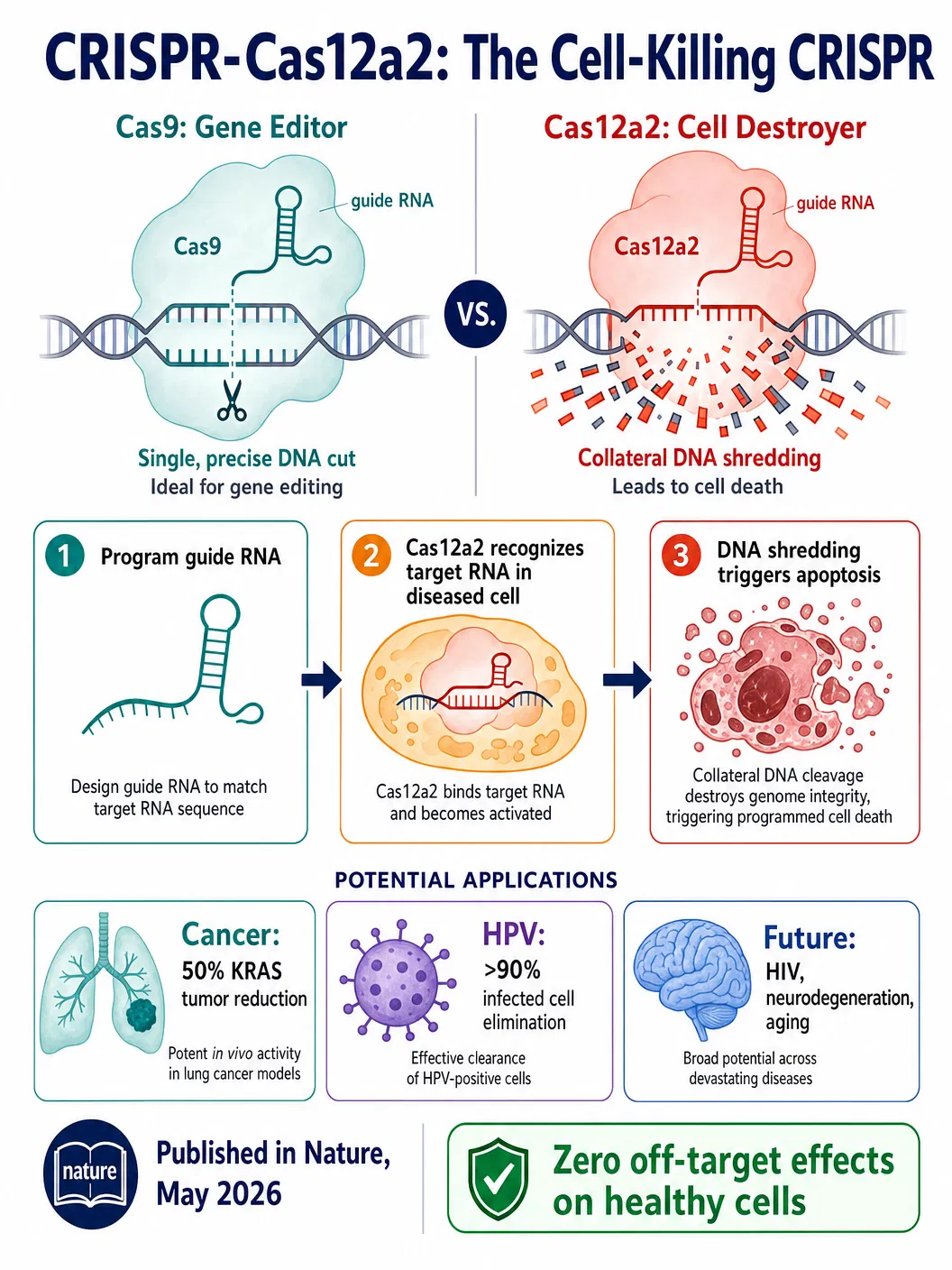
For more than a decade, CRISPR has been synonymous with gene editing — the precise molecular scissors that cut DNA at a specific location to correct a mutation, insert a gene, or disable a faulty sequence. The technology earned its pioneers a Nobel Prize and has already produced approved therapies for sickle cell disease and transfusion-dependent beta-thalassaemia. But on 6 May 2026, researchers at the University of Utah Health, Utah State University, the Helmholtz Institute for RNA-based Infection Research, and Akribion Therapeutics published a study in Nature that introduces an entirely different application of CRISPR biology — one that does not edit genes at all. Instead, it kills cells.
The technology centres on Cas12a2, a protein distantly related to the Cas9 enzyme used in conventional CRISPR editing. Like Cas9, Cas12a2 can be programmed with a short guide RNA to recognise a specific genetic sequence. But the similarity ends there. When Cas9 finds its target, it makes a single precise cut in the DNA double helix — a surgical incision that the cell’s own repair machinery can then exploit to introduce a desired change. When Cas12a2 finds its target, it begins cutting DNA indiscriminately and does not stop. The result is catastrophic genomic damage that triggers the cell to self-destruct through a process called apoptosis.
“Its goal is not to correct anything,” explained Dr. Yang Liu, assistant professor in biochemistry at the University of Utah Health and co-senior author of the study. “Instead, it’s to destroy anything it sees.”
This might sound more dangerous than therapeutic. A molecule that shreds DNA could, in theory, cause widespread tissue destruction. But the critical insight of the study is that Cas12a2 is activated not by DNA but by RNA — the intermediate molecule that cells produce when they read their genes. This means Cas12a2 can be programmed to recognise RNA sequences that are unique to diseased cells, such as those produced by cancer-driving mutations or viral infections, while remaining completely inert in healthy cells that do not produce those specific RNA molecules.
The researchers demonstrated this principle in two compelling disease models. First, they targeted Cas12a2 to a cancer-causing mutation in the KRAS gene — the same oncogene that drives pancreatic, lung, and colorectal cancers. In human lung cancer cells carrying the KRAS mutation, Cas12a2 reduced cell growth by approximately fifty percent — comparable to the efficacy of established chemotherapy drugs like cisplatin. But unlike cisplatin, which poisons dividing cells indiscriminately and causes devastating side effects, Cas12a2 did not affect healthy cells with normal KRAS at all.
“The enzyme that we’re working with is extremely specific,” Dr. Liu said. “It does not touch the healthy cells. So if we’re thinking about a cancer therapy, you’re treating cancer with no side effects. That was striking to us. We did not know that was possible.”
Second, the team targeted Cas12a2 to RNA produced by human papillomavirus (HPV), the virus responsible for cervical cancer, genital warts, and several other malignancies. Collaborators at Akribion Therapeutics found that Cas12a2 reduced the growth of HPV-infected cells in laboratory dishes by more than ninety percent without harming uninfected cells. When injected directly into HPV-driven tumours in mice, the treatment slowed tumour growth significantly — demonstrating that the approach can work in living organisms, not just in isolated cells.
“Our technology provides us with a powerful tool for sequence-specific depletion of pathogenic cells,” said Dr. Paul Scholz, co-founder and head of research and development at Akribion Therapeutics and co-first author on the study.
The implications extend far beyond cancer and HPV. Because Cas12a2 can be programmed to recognise virtually any RNA sequence, the researchers believe it could be directed against cells infected by HIV, hepatitis B, Epstein-Barr virus, or any other pathogen that produces distinctive RNA transcripts. Dr. Liu also envisions applications in neurodegenerative diseases, where Cas12a2 could eliminate brain cells that produce toxic proteins, and in aging-related conditions, where it could clear senescent cells that consume resources without contributing function.
The path to clinical application remains long. The vast majority of experiments have been conducted in cells in laboratory dishes, and significant work is needed to test safety and efficacy in animal models before human trials become feasible. Delivering sufficient quantities of Cas12a2 to the right tissues in the body will be a fundamental engineering challenge. But the researchers are optimistic about the technology’s transformative potential.
“Because Cas12a2 can be programmed with a guide RNA to target any RNA sequence, and it shows little to no off-targeting, we believe we have discovered a way to selectively kill cells across all of biology,” said Dr. Ryan Jackson, associate professor of chemistry and biochemistry at Utah State University and co-senior author. “We envision this technology will transform science, agriculture, and medicine in ways previously unavailable.”
When asked what comes next, Dr. Liu’s answer was characteristically ambitious: “Curing the incurables.”
References: Thompson J, Liu Y, Jackson R, Beisel C, Scholz P, et al. RNA-triggered cell killing with CRISPR-Cas12a2. Nature. 6 May 2026. University of Utah Health News Release, 6 May 2026. Akribion Therapeutics.