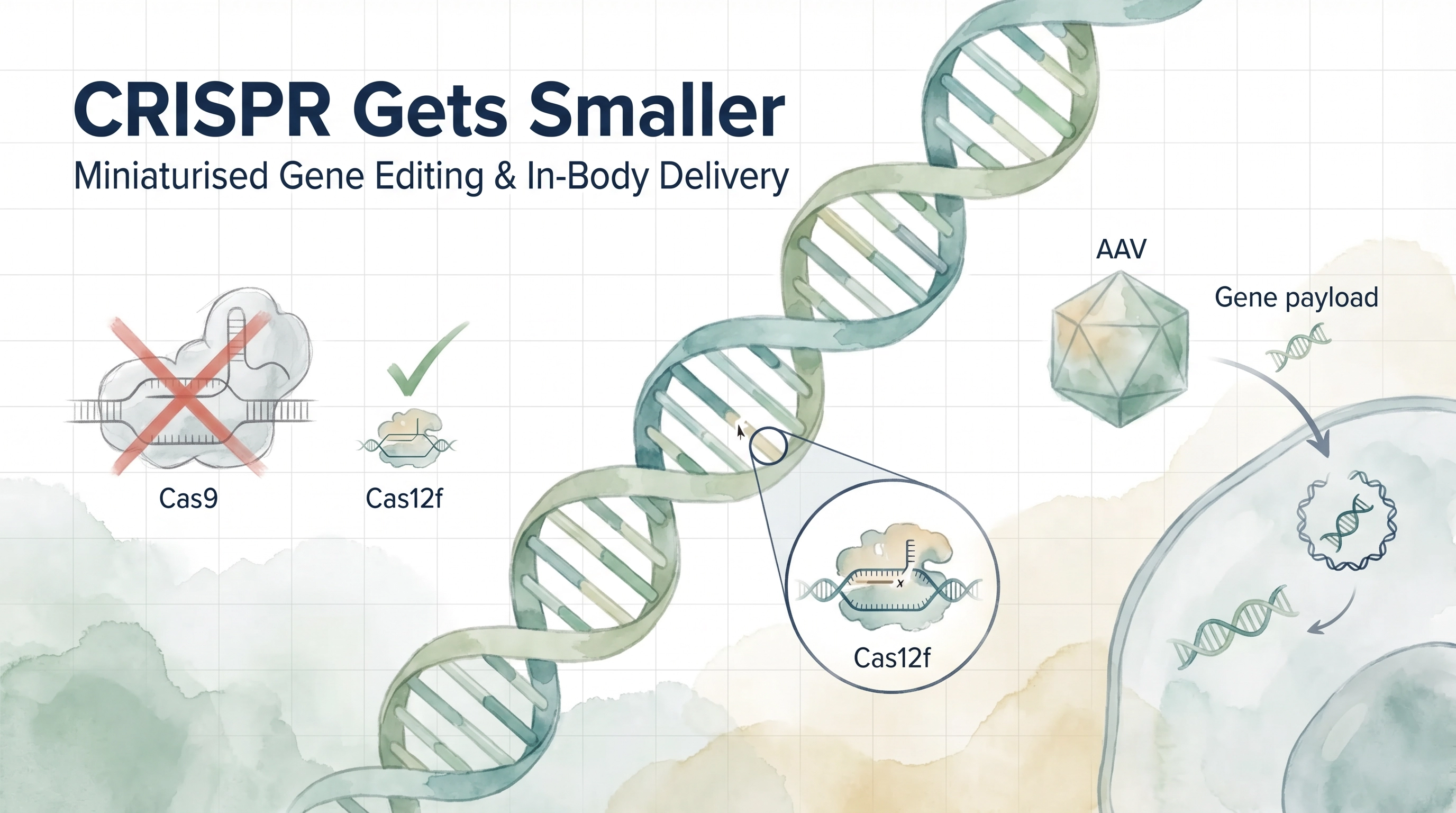
Since Jennifer Doudna and Emmanuelle Charpentier received the 2020 Nobel Prize in Chemistry for developing CRISPR-Cas9, the gene-editing field has faced a persistent engineering paradox: the most powerful editing proteins are too large to be delivered precisely inside the living human body. The dominant delivery vehicle for gene therapies — adeno-associated virus (AAV) vectors — has a strict cargo capacity, and the workhorse Cas9 enzyme simply does not fit. As a result, most approved CRISPR-based therapies have been limited to ex vivo approaches, where cells are extracted from a patient, edited in the laboratory, and reinfused. Effective, but slow, expensive, and inaccessible to the vast majority of patients worldwide.
On 13 April 2026, the National Institutes of Health announced a breakthrough that may resolve this constraint entirely.
The Al3Cas12f Discovery
Researchers at the University of Texas at Austin, supported by NIH's National Institute of General Medical Sciences, identified a naturally occurring enzyme called Al3Cas12f — a Cas12f-family protein small enough to fit within AAV vectors. Using a combination of cryo-electron microscopy imaging and machine learning structural analysis, the team characterised why this enzyme outperforms other compact Cas proteins: it forms a more stable, tightly connected protein-RNA complex that arrives essentially pre-assembled and ready to function in human cells.
The team then engineered an enhanced variant, Al3Cas12f RKK, which elevated gene-editing efficiency from below 10 percent to more than 80 percent across a range of tested genomic targets. In one commonly edited region of the genome, efficiency reached 90 percent — a figure that rivals or exceeds the performance of much larger editing systems. Critically, the mutations targeted in these experiments were associated with cancer, atherosclerosis, and amyotrophic lateral sclerosis (ALS), three conditions for which in-body gene correction has long been a therapeutic aspiration rather than a clinical reality.
"Smart delivery of gene editing systems is a powerful notion with broad clinical implications, and this basic science finding takes us a significant step toward that future." — Erica Brown, Ph.D., Acting Director, NIH National Institute of General Medical Sciences
The research was published in Nature Structural & Molecular Biology (DOI: 10.1038/s41594-026-01788-6).
A Field in Full Bloom
The Al3Cas12f RKK result arrives in the context of a gene therapy field experiencing its most productive period on record. The 2026 Breakthrough Prize in Life Sciences was awarded to researchers whose work produced approved gene therapies for sickle cell disease and inherited blindness — conditions that had resisted conventional treatment for generations. The FDA granted its first cell and gene therapy approval of 2026 to KRESLADI, Rocket Pharma's treatment for severe Leukocyte Adhesion Deficiency-I, a rare and often fatal immune disorder. Duke Eye Center became the first US academic medical centre to implant a cell-based gene therapy for macular degeneration.
Meanwhile, the frontier of CRISPR technology is advancing beyond simple gene disruption. Base editing and prime editing — techniques pioneered in the laboratory of David Liu at the Broad Institute — allow scientists to make precise, single-letter corrections to the DNA code without cutting both strands of the double helix, dramatically reducing the risk of unintended mutations. A preprint published in April 2026 demonstrated cardiac prime editing capable of rescuing cardiomyopathy in animal models, while researchers reported improving integration of the XIST gene into the extra chromosome 21 in Down syndrome models by approximately 30-fold over conventional CRISPR approaches.
What Comes Next
The UT Austin team's immediate next step is to package Al3Cas12f RKK into actual AAV vectors and test its performance in vivo — the critical bridge between cell-culture results and clinical translation. If those experiments succeed, the implications extend far beyond the three diseases already targeted. Any condition with a defined genetic driver and an accessible tissue target becomes, in principle, a candidate for in-body CRISPR correction: certain forms of inherited blindness, muscular dystrophy, familial hypercholesterolaemia, and a range of rare monogenic disorders.
The miniaturisation of CRISPR is not merely a technical milestone. It is the key that could unlock gene therapy for the many rather than the few.
Sources: NIH News Release — NIH-funded breakthrough shrinks CRISPR for precision delivery in the body (April 13, 2026); The Scientist — Gene Therapy Pioneers Win 2026 Breakthrough Prize (April 18, 2026); Reuters — Researchers eye potential Down syndrome fix via advanced gene editing (April 17, 2026); MedReport Foundation — Base Editing vs. Prime Editing (April 2026).