Pancreatic cancer has long occupied a uniquely devastating position in oncology. With a five-year survival rate hovering below ten percent and most patients diagnosed only after the disease has metastasised beyond surgical reach, it has resisted nearly every therapeutic advance that has transformed outcomes in other cancers. The primary treatment remains chemotherapy — a blunt instrument that offers modest benefit in the second-line setting and exacts a heavy toll on quality of life. But on 6 May 2026, a publication in the New England Journal of Medicine signalled that the landscape may be shifting in a fundamental way.
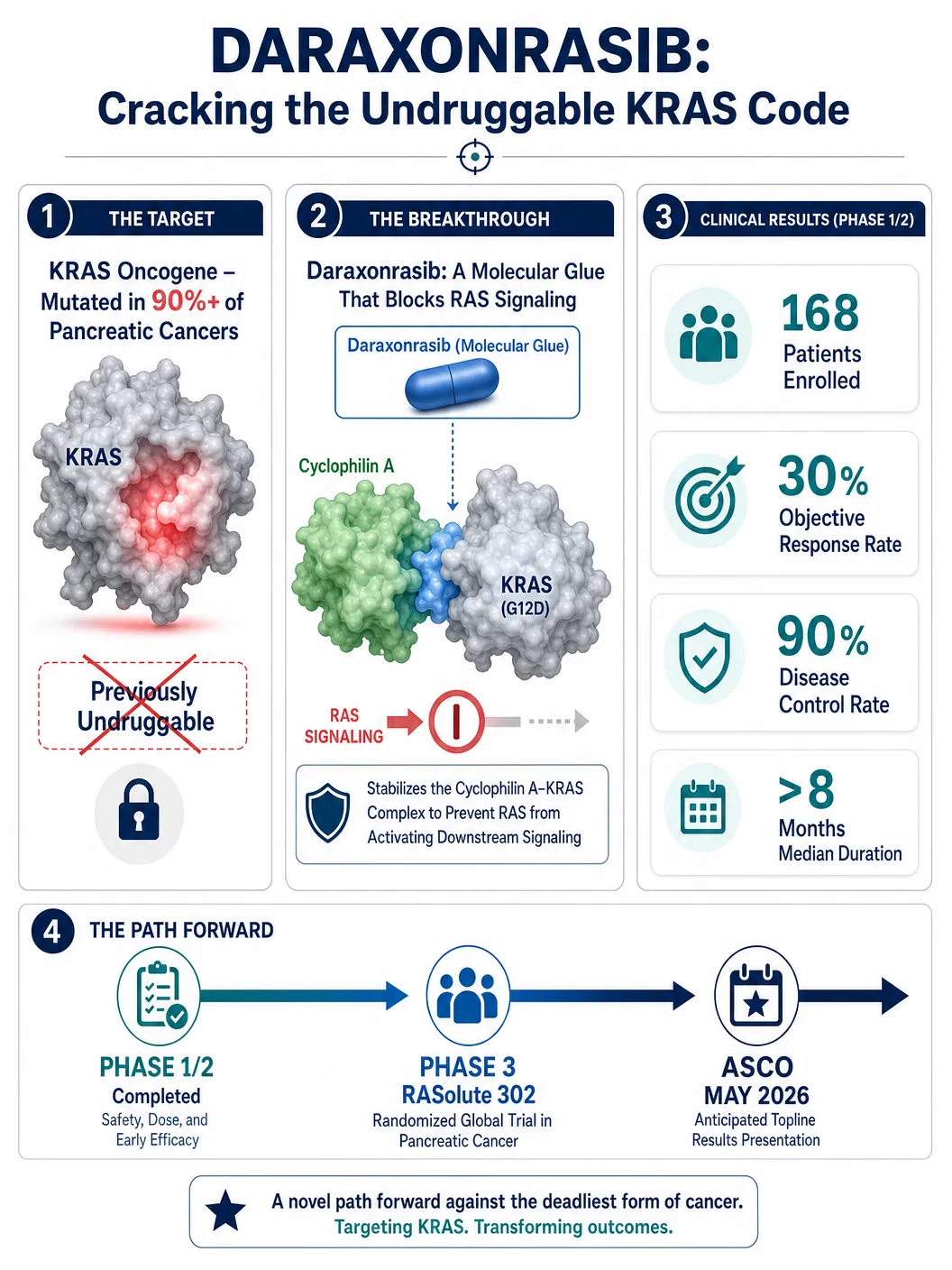
The study, led by Dr. Brian Wolpin at Dana-Farber Cancer Institute’s Hale Family Center for Pancreatic Cancer Research, reported results from a first-in-human phase 1/2 trial of daraxonrasib — a RAS(ON) multi-selective inhibitor that targets the very oncogene driving more than ninety percent of pancreatic cancers. For decades, the KRAS protein was considered “undruggable,” a molecular target so structurally smooth and featureless that no small molecule could gain purchase on its surface. That dogma has now been decisively overturned.
Daraxonrasib works through an elegant mechanism: it functions as a molecular glue that recruits cyclophilin A, a naturally occurring cellular protein, to physically block the signalling output of mutant RAS proteins. Unlike earlier RAS inhibitors — two of which are now approved for lung and colorectal cancers — daraxonrasib is active against the specific KRAS mutations that predominate in pancreatic cancer, particularly KRAS G12D and G12V. This distinction is critical: the approved drugs target KRAS G12C, a mutation found in only one to two percent of pancreatic tumours.
The phase 1/2 trial enrolled 168 patients with advanced, RAS-mutant pancreatic cancer who had already failed at least one line of chemotherapy. At the recommended phase 2 dose of 300 milligrams daily — administered as a simple oral pill — the results were striking. Approximately thirty percent of patients with one prior line of therapy achieved an objective tumour response, meaning their cancers measurably shrank. Even more remarkably, roughly ninety percent of all patients experienced disease control, defined as tumour shrinkage or stabilisation. The median duration of response exceeded eight months for patients with one prior therapy — substantially longer than what second-line chemotherapy typically achieves.
The safety profile was manageable. The most common adverse events were rash, oral inflammation, nausea, and diarrhoea — side effects that were generally controlled with supportive medications. No unexpected toxicities emerged, and the drug’s oral formulation represents a significant quality-of-life advantage over intravenous chemotherapy regimens.
“If supported by data from future clinical trials, daraxonrasib would be a targeted therapy relevant to nearly all patients with advanced pancreatic cancer,” Dr. Wolpin stated. “This trial provides the first published data showing the safety and broad activity of a RAS(ON) multi-selective inhibitor in pancreatic cancer. If this drug proves effective in larger clinical trials, it would signify a substantial shift in how this disease is treated.”
The phase 3 confirmatory trial, RASolute 302, is already underway and will directly compare daraxonrasib to standard second-line chemotherapy. Primary results are expected at the American Society for Clinical Oncology (ASCO) Annual Meeting on 31 May 2026 — a plenary presentation that reflects the oncology community’s recognition of the study’s potential significance.
The broader implications extend beyond a single drug. Daraxonrasib is the first of what is expected to be a wave of RAS-targeting therapies for pancreatic cancer. Multiple pharmaceutical companies are now advancing their own KRAS inhibitors through clinical development, creating a competitive landscape that could accelerate progress and ultimately offer patients combination strategies that attack the disease from multiple angles simultaneously.
For the estimated 64,000 Americans who will be diagnosed with pancreatic cancer in 2026 — and the hundreds of thousands worldwide — this represents something that has been vanishingly rare in the history of this disease: genuine, evidence-based hope.
References: Wolpin BM, et al. Daraxonrasib in RAS-Mutant Metastatic Pancreatic Cancer: A Phase 1/2 Trial. New England Journal of Medicine. 2026. Dana-Farber Cancer Institute News Release, 6 May 2026. MD Anderson Cancer Center Research Newsroom, 6 May 2026. Revolution Medicines Clinical Data.