Pancreatic cancer carries one of the most sobering statistics in all of oncology: a five-year survival rate of just 13 percent. Even when surgeons successfully remove a tumour, cancer cells frequently hide in the body and re-emerge months or years later. A landmark study published this week in Developmental Cell by researchers at the University of Rochester Medicine's Wilmot Cancer Institute has uncovered a key molecular mechanism behind this notorious resilience — and it involves the body's own internal clock.
The Dec2 Gene: A Molecular Disguise
The research, led by surgeon-scientist Darren Carpizo, MD, PhD, centres on a gene called Dec2 (also known as BHLHE41). Dec2 was previously known for its role in regulating sleep duration — rare variants of the gene are associated with people who function well on fewer than six hours of sleep. But Carpizo's team has now revealed an entirely new function: Dec2 can disguise pancreatic cancer cells from the immune system's killer T-cells.
The mechanism works through a surface molecule on tumour cells. When Dec2 is active, it regulates the expression of this surface molecule in a way that effectively renders the cancer cell invisible to patrolling T-cells. The immune system's sentinels pass by without triggering an attack.
"Pancreatic cancer is an urgent problem. Our new study brings us another step closer to understanding how these pancreas tumour cells can hide out for long periods of time, and how to target them." — Darren Carpizo, MD, PhD, Wilmot Cancer Institute
When the researchers knocked out Dec2 in laboratory models, the results were striking: immune cells were suddenly able to find and engage the pancreatic cancer cells. This finding positions Dec2 as a potential new therapeutic target — a molecular lock that, if opened, could expose hidden tumour cells to immune attack.
The Circadian Dimension
The discovery takes on an additional layer of complexity because Dec2 does not operate at a constant level. Like many genes involved in circadian biology, its expression rises and falls across a 24-hour cycle. Dec2 levels within pancreatic cancer cells go up during the day and down at night, creating windows of greater and lesser immune evasion.
This rhythmic behaviour provides a biological explanation for a clinical observation that has puzzled oncologists for years: immunotherapy drugs appear to work better when administered in the morning than in the evening. The circadian oscillation of Dec2 — and the corresponding fluctuation in tumour cell visibility — may be the underlying reason.
| Time of Day | Dec2 Expression | T-cell Recognition | Immunotherapy Efficacy |
|---|---|---|---|
| Morning (6–10 am) | Rising | Reduced evasion | Higher |
| Midday (12–2 pm) | Peak | Maximum evasion | Lower |
| Evening (6–10 pm) | Falling | Partial evasion | Moderate |
| Night (12–4 am) | Trough | Minimum evasion | Potentially highest |
Table 1: Inferred relationship between Dec2 circadian expression and T-cell recognition windows. Clinical validation is ongoing.
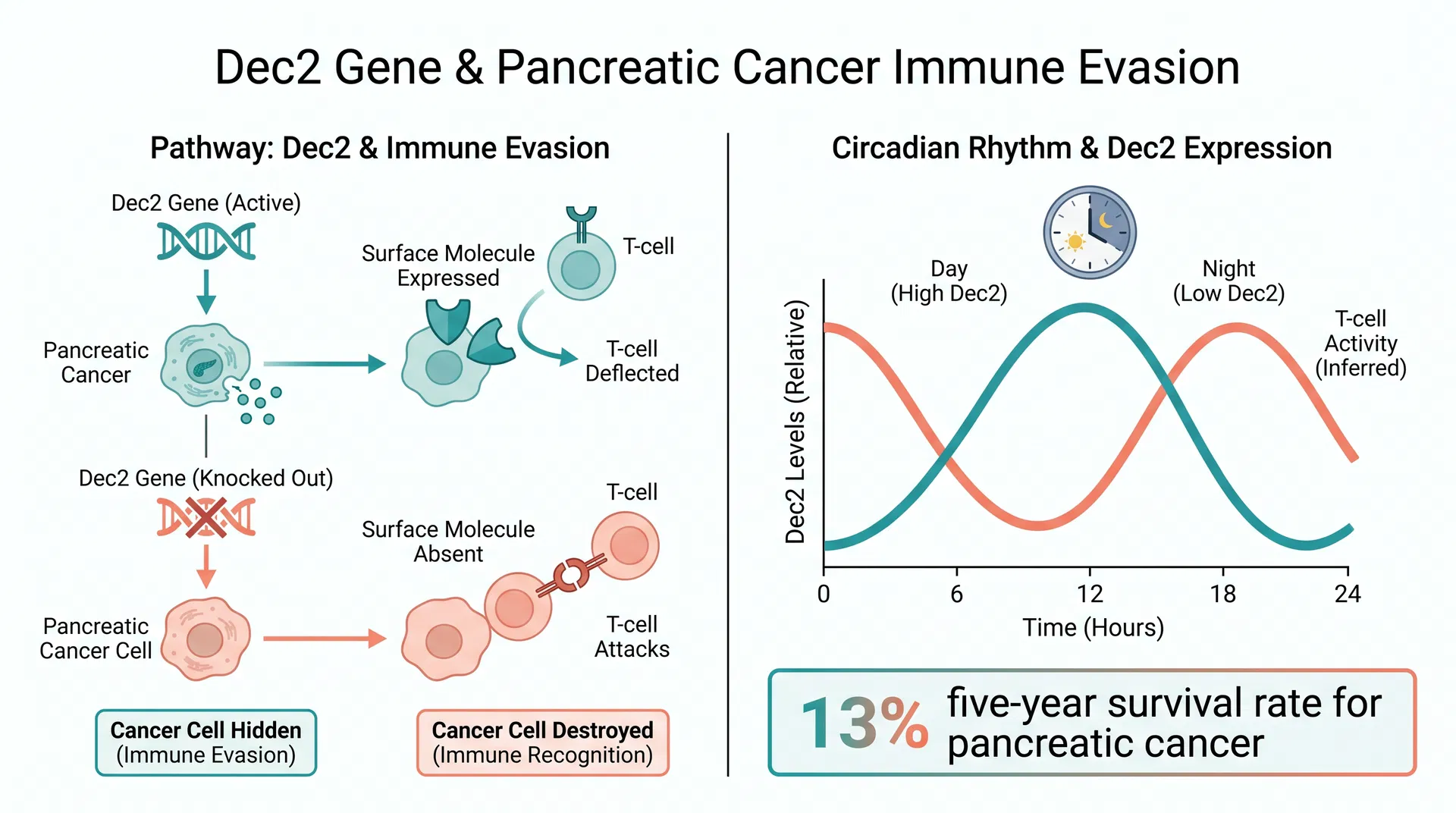
Figure 1: Left — Dec2 active state shields cancer cells from T-cell attack; Dec2 knockout restores immune recognition. Right — Dec2 expression oscillates over 24 hours, inversely correlating with T-cell activity windows.
Implications for the mRNA Pancreatic Cancer Vaccine
The Dec2 discovery has immediate relevance for one of the most exciting developments in pancreatic cancer research: the experimental mRNA vaccine being tested at Memorial Sloan Kettering. In a small clinical trial of 16 patients, the vaccine boosted survivorship in eight participants — but the other eight did not respond.
Vaccines of this type depend entirely on T-cells being able to seek out and destroy cancer cells. If Dec2 activity is preventing T-cells from recognising tumour cells in non-responders, targeting Dec2 could be the key to converting vaccine non-responders into responders.
"Our research has important implications for this, because if the actions of Dec2 will not allow the vaccine to work properly, it may explain why 50 percent of the patients didn't do well," Carpizo said. "Targeting Dec2 may be an alternative solution."
Building on a Foundation of Circadian Cancer Biology
This is not the first circadian gene Carpizo's team has linked to pancreatic cancer. Their earlier work, funded by a pilot grant from Wilmot in 2023, identified BMAL1 — another core circadian clock gene — as a regulator of pancreatic cancer progression and metastasis through immune evasion. The National Cancer Institute has since supported the expanded research programme.
The convergence of two separate circadian genes in pancreatic immune evasion suggests that the tumour's exploitation of the body's internal clock is not incidental but deeply embedded in its survival strategy. Future research will need to determine whether chronotherapy — the deliberate timing of drug administration to align with circadian biology — can meaningfully improve outcomes for patients with pancreatic cancer.
What Comes Next
Carpizo's laboratory is now working to identify the specific surface molecule regulated by Dec2, which would provide a direct drug target. The team is also designing studies to test whether Dec2 inhibition, combined with existing immunotherapy agents, can produce synergistic tumour killing in their genetically engineered mouse model.
For the 60,000 Americans diagnosed with pancreatic cancer each year, a therapy that strips away the molecular disguise enabling immune evasion could represent a fundamental shift in the treatment landscape — one where the cancer's own clock becomes its greatest vulnerability.
Source: University of Rochester Medicine / Wilmot Cancer Institute, published in Developmental Cell, May 2026. Research supported by the National Cancer Institute.