Gene drives represent one of the most powerful and controversial tools in modern biology. By harnessing the mechanisms of inheritance, they can spread engineered genetic traits through wild populations with a speed and completeness that no previous biotechnology has approached. A single release of gene-drive-carrying organisms could, in theory, alter or eliminate an entire species across a continent within a few generations. The implications — for disease control, conservation, agriculture, and biosecurity — are profound. So, too, are the governance challenges.
The Biology of Gene Drives
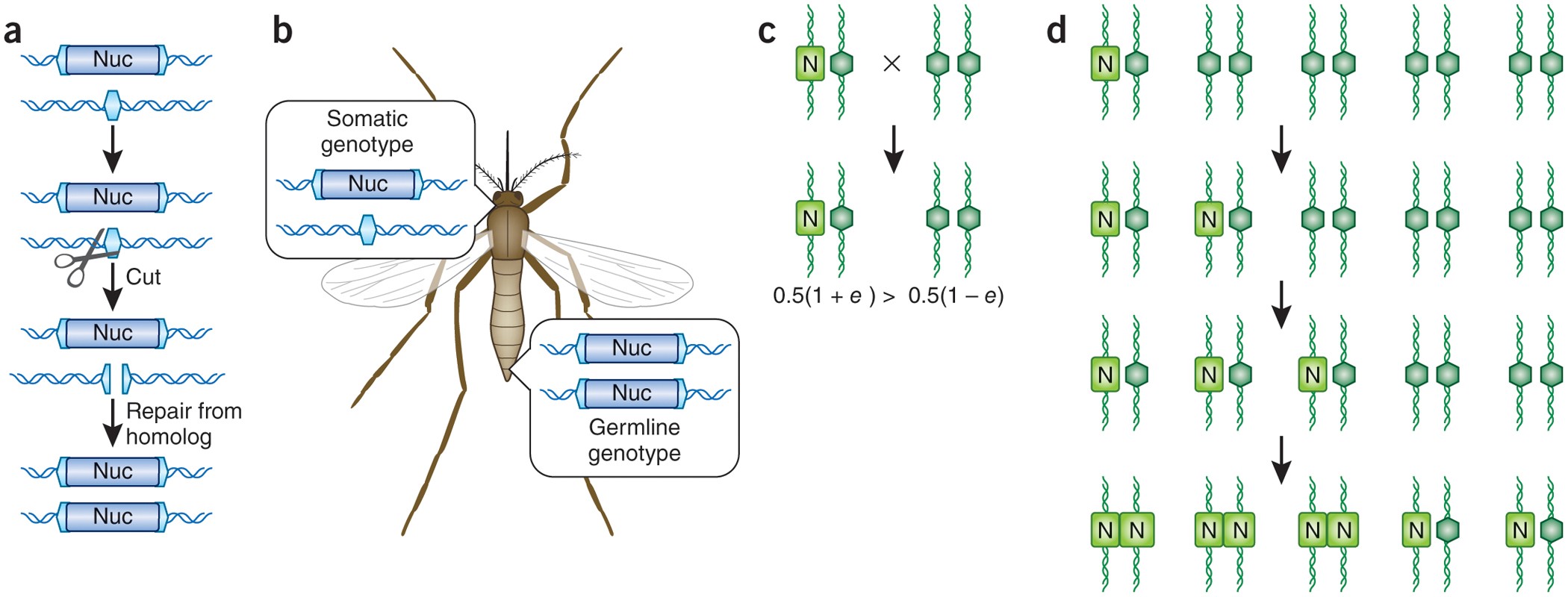
In natural populations, a gene inherited from one parent has, on average, a 50% chance of being passed to offspring. Gene drives subvert this Mendelian rule through a process called "super-Mendelian inheritance" — engineering a genetic element that copies itself into both chromosomes of every organism that carries it, ensuring it is passed to nearly 100% of offspring rather than 50%. Over successive generations, a gene drive can spread through an entire population even if it confers no fitness advantage — or even a modest fitness cost.
The mechanism most commonly used in contemporary gene drive research is CRISPR-Cas9. A CRISPR gene drive construct encodes both the guide RNA that directs the Cas9 enzyme to a specific genomic target and the Cas9 enzyme itself, along with the desired payload gene. When the construct is inserted into one chromosome, the Cas9 enzyme cuts the corresponding site on the other chromosome, and the cell's repair machinery copies the drive construct into the cut site. The result is an organism that is homozygous for the drive — and that will pass it to essentially all of its offspring.
Potential Applications
The most widely discussed application of gene drives is in the control of vector-borne diseases. Malaria kills over 600,000 people annually, the vast majority of them children in sub-Saharan Africa. The primary vector, Anopheles gambiae, is the target of two complementary gene drive strategies: population suppression drives, which spread a fertility-impairing gene through female mosquitoes, reducing population size; and population replacement drives, which spread genes conferring resistance to the malaria parasite, leaving the mosquito population intact but unable to transmit disease. Field trials of both approaches are advancing, with the Target Malaria consortium leading work in West Africa.
Beyond disease control, gene drives are being explored for conservation applications — specifically, the control of invasive species that are driving native species to extinction. In New Zealand, the "Predator Free 2050" initiative has explored whether gene drives could be used to suppress populations of invasive rats, stoats, and possums that have devastated native bird populations. In the Galápagos, similar discussions are underway regarding invasive rodents that threaten the iconic fauna of the archipelago.
Agricultural applications include the suppression of crop pests and the potential elimination of herbicide-resistant weeds. In each case, the appeal of gene drives lies in their capacity for self-propagating, population-wide effect — a single intervention that spreads without repeated application.
Biosafety and Biosecurity Risks
The same properties that make gene drives potentially transformative also make them potentially dangerous. A gene drive released into a wild population cannot be recalled. If it spreads beyond its intended target species — through hybridisation with related species, or through the accidental release of organisms intended only for contained laboratory study — the consequences could be irreversible. The extinction of a species, even an invasive or disease-carrying one, can have cascading ecological effects that are difficult to predict and impossible to undo.
The biosecurity dimensions are equally serious. Gene drives represent a potential dual-use technology: the same capability that could suppress a disease vector could, in principle, be weaponised to suppress a beneficial species, introduce a harmful trait into a wild population, or drive a species to extinction as an act of ecological warfare. The 2016 US National Academies of Sciences report on gene drives noted that the technology "poses risks that are difficult to assess" and called for a robust international governance framework — a call that has yet to be fully answered.
Containment strategies are an active area of research. "Daisy chain" drives, which require multiple interacting genetic elements to propagate, offer a potential mechanism for geographic or temporal limitation. "Reversal drives" that can overwrite a previously released drive have been demonstrated in laboratory settings. But the reliability of these safeguards in open-field conditions, across diverse genetic backgrounds and ecological contexts, remains to be established.
The Governance Landscape
The governance of gene drives is one of the most complex challenges in contemporary biosafety policy. It sits at the intersection of national sovereignty, international environmental law, indigenous rights, and the ethics of irreversible interventions. The Convention on Biological Diversity (CBD) has been the primary international forum for gene drive governance discussions, with the Cartagena Protocol on Biosafety providing the most relevant existing framework — though one that was not designed with self-propagating genetic elements in mind.
Key governance questions include: who has the authority to approve the release of a gene drive? How should affected communities — including those in countries where a drive might spread without deliberate release — be consulted and their consent obtained? What liability frameworks apply if a drive causes unintended harm? How should the international community respond to unilateral releases by states or non-state actors?
The development of adequate answers to these questions requires not only scientific expertise but also deep engagement with ethics, law, political science, and the communities most directly affected. It is a governance challenge of the first order — and one that the biosafety community must engage with urgency, before the technology outpaces the frameworks designed to govern it.
Conclusion
Gene drives are neither a panacea nor a catastrophe waiting to happen — they are a powerful technology whose consequences depend entirely on how wisely they are developed and governed. The scientific community has a responsibility to be transparent about both the potential and the risks, to invest in containment and reversal strategies, and to engage proactively with the governance processes that will determine how and whether this technology is deployed. The stakes — for human health, for biodiversity, and for the integrity of the biosphere — could hardly be higher.