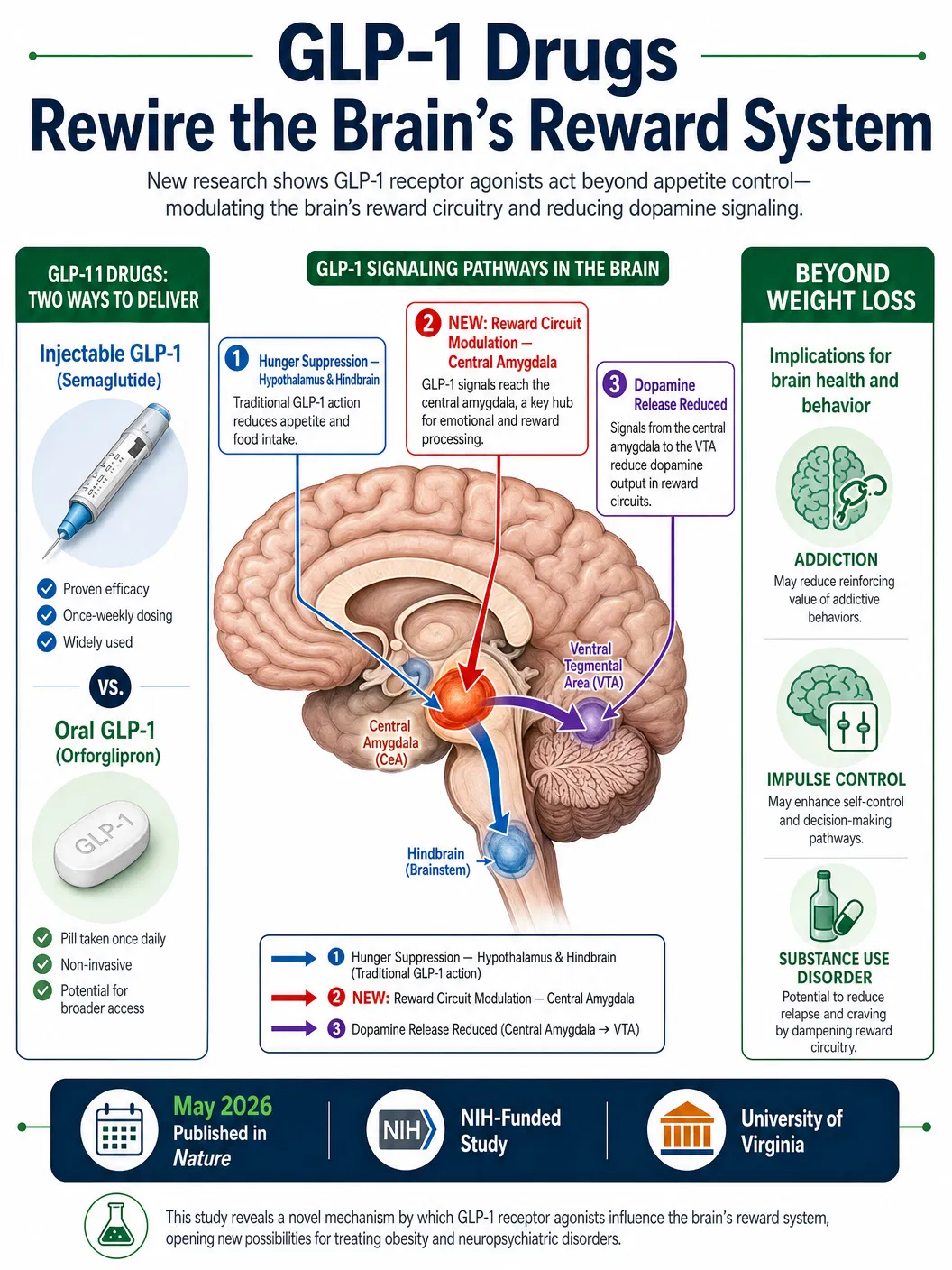
The global conversation about weight-loss drugs has entered a fundamentally new chapter. On 6 May 2026, the National Institutes of Health announced the results of a landmark study, published in Nature, demonstrating that an emerging class of oral small-molecule GLP-1 receptor agonists does far more than suppress appetite — these drugs penetrate deep into the brain and directly modulate the reward circuitry that governs pleasure-seeking behaviour, hedonic feeding, and potentially addiction itself.
The study, conducted by researchers at the University of Virginia and funded by the NIH's National Institute on Drug Abuse, specifically investigated orforglipron and danuglipron — next-generation GLP-1 drugs that can be taken as a daily pill rather than injected. Unlike their injectable predecessors such as semaglutide (marketed as Ozempic and Wegovy), which primarily suppress hunger-driven eating through hypothalamic and hindbrain circuits, these oral compounds engage an entirely separate neural pathway that scientists had not previously associated with GLP-1 signalling.
The research team, led by neuroscientist Ali D. Güler, modified the GLP-1 receptors of mice using gene-editing techniques to make them more humanlike, then administered the oral drugs and mapped their activity throughout the brain. The results were striking: while the drugs activated familiar appetite-suppression regions, they also triggered robust activity in the central amygdala — a brain structure associated with desire, emotional processing, and reward valuation that lies deeper in the brain than scientists previously believed GLP-1 drugs could directly reach.
"We've known that GLP-1 drugs suppress feeding behaviour driven by energy demand," Güler explained in the university's announcement. "Now it seems oral small-molecule GLP-1s also dial back eating for pleasure by engaging a brain reward circuit." In more vivid terms, he described the mechanism: "They're acting on the system that makes you want the cake, not just the system that makes you feel full."
The implications extend far beyond weight management. Once activated by these drugs, neurons in the central amygdala reduced dopamine release into key hubs of the brain's reward circuitry during hedonic feeding — the same dopamine pathways implicated in substance use disorders, gambling addiction, and compulsive behaviours. Lorenzo Leggio, clinical director of NIH's National Institute on Drug Abuse, emphasised the urgency of understanding these mechanisms: "As the accessibility of these medications continues to rise and patient uptake increases, it's crucial that we understand the neural mechanisms underlying the effects we're seeing."
The timing of this discovery coincides with a dramatic expansion in GLP-1 drug accessibility. On the same day the Nature study was published, the Centers for Medicare and Medicaid Services announced that beginning July 2026, Medicare beneficiaries will have access to GLP-1 medications for just fifty dollars per month — a policy decision that will bring these drugs to millions of Americans who previously could not afford them. The convergence of scientific understanding and policy action suggests that GLP-1 drugs are entering a new era of both clinical application and public health impact.
The distinction between hunger suppression and reward modulation is not merely academic — it has profound clinical significance. Many patients who take GLP-1 drugs report not just reduced appetite but a fundamental shift in their relationship with food, alcohol, and other pleasurable substances. Anecdotal reports of reduced alcohol consumption, decreased interest in shopping, and diminished gambling urges among GLP-1 users have circulated for years, but until now lacked a clear neurobiological explanation. The discovery of the central amygdala pathway provides exactly that explanation: these drugs are not simply making people feel full, they are recalibrating the brain's valuation of rewarding experiences.
The oral formulation represents a critical advance in accessibility and patient compliance. Injectable GLP-1 drugs require weekly subcutaneous injections, cold-chain storage, and carry a significant cost burden. Oral small-molecule compounds like orforglipron — already FDA-approved — can be manufactured more cheaply, stored at room temperature, and taken as a simple daily pill. The Nature study demonstrates that these oral drugs achieve their effects through a distinct mechanism from their injectable counterparts, suggesting that the two classes may eventually be used in complementary therapeutic strategies.
The research team is now planning follow-up studies to investigate whether oral GLP-1 drugs can specifically reduce cravings associated with substance use disorder. If the central amygdala pathway proves to be therapeutically accessible for addiction treatment, it could represent one of the most significant pharmacological advances in addiction medicine in decades — transforming a class of drugs originally developed for diabetes into a broad-spectrum intervention for disorders of reward processing.
For the broader field of neuroscience, the study challenges long-held assumptions about the blood-brain barrier's selectivity and the depth to which small-molecule drugs can penetrate neural tissue. The finding that oral GLP-1 compounds reach and activate the central amygdala — a structure buried deep within the temporal lobe — opens new questions about what other brain regions these drugs may influence and what additional therapeutic applications may emerge as researchers continue to map their neural effects.
References: Godschall EN, Gungul TB, Sajonia IR, et al. A Brain Reward Circuit Inhibited By Next-Generation Weight Loss Drugs in Mice. Nature. 2026. DOI: 10.1038/s41586-026-10444-4. NIH News Release, 6 May 2026. University of Virginia College of Arts & Sciences, 6 May 2026. CMS Press Release, 6 May 2026.