The Gut-Brain Connection Gets Molecular
For years, researchers have observed a statistical association between the gut microbiome and mental health. Studies have linked altered gut bacterial communities to depression, anxiety, and cognitive decline. But correlation is not mechanism, and the field has struggled to answer the fundamental question: how, precisely, do bacteria in the intestine influence the brain?
A study published in the Journal of the American Chemical Society on 25 April 2026 by researchers at Harvard Medical School has provided the clearest molecular answer yet — and it involves an unexpected environmental contaminant acting as a biochemical trigger.
The Bacterium Under Investigation
Morganella morganii is a gram-negative bacterium found in the human gut. It is not a pathogen in the traditional sense — it is a commensal organism present in many healthy individuals. But several independent studies have linked elevated M. morganii abundance to major depressive disorder (MDD), as well as to inflammatory conditions including type 2 diabetes and inflammatory bowel disease.
The question was whether M. morganii contributes to depression, whether depression changes the microbiome (reverse causation), or whether a third factor explains both. The Harvard team set out to find the molecular mechanism that would distinguish these possibilities.
The Unexpected Culprit: An Environmental Contaminant
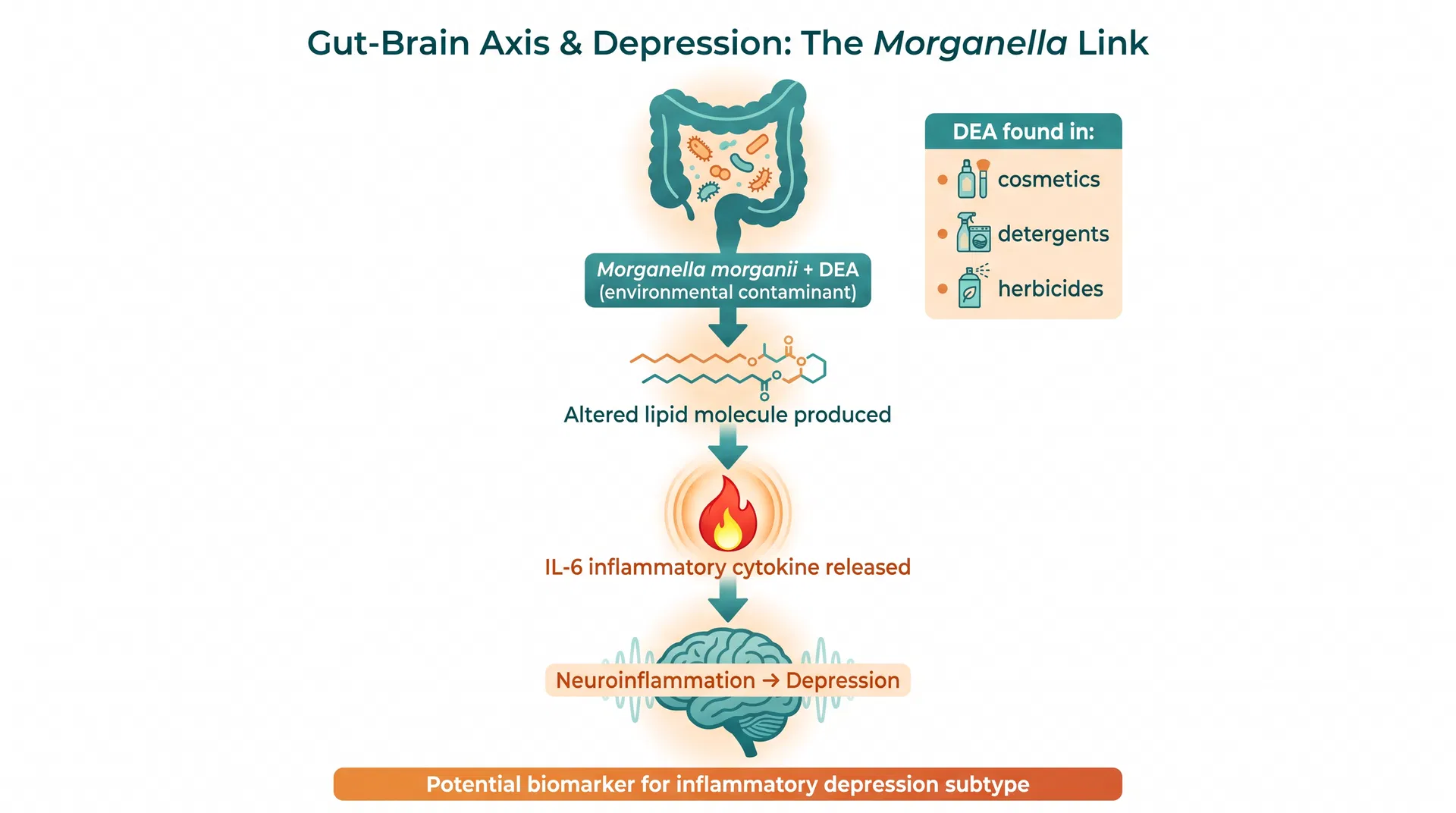
The researchers discovered that an environmental contaminant called diethanolamine (DEA) — commonly found in industrial, agricultural, and consumer products including cosmetics, detergents, and herbicides — can infiltrate the biochemistry of M. morganii in a specific and consequential way.
In normal circumstances, M. morganii produces a lipid molecule containing a sugar alcohol. DEA, when present in the gut environment, can substitute for that sugar alcohol, producing an altered lipid molecule with dramatically different biological properties.
The normal version of the molecule is essentially inert. The DEA-substituted version is not. It activates the immune system, prompting the release of inflammatory cytokines — specifically interleukin-6 (IL-6).
"We knew that micropollutants can be incorporated into fatty molecules in the body, but we didn't know how this occurs or what happens next. DEA's metabolism into an immune signal was completely unexpected." — Jon Clardy, Harvard Medical School
The Inflammation-Depression Connection
The significance of IL-6 in this context cannot be overstated. Chronic elevation of IL-6 is one of the most consistently replicated biological findings in major depressive disorder. Multiple meta-analyses have confirmed that depressed patients have significantly higher circulating IL-6 levels than healthy controls, and that successful antidepressant treatment is associated with IL-6 normalisation.
The Harvard finding provides a plausible upstream mechanism: environmental DEA → M. morganii biochemical alteration → DEA-substituted lipid → IL-6 release → neuroinflammation → depression.
This is not a complete causal chain — more research is needed to confirm each step in humans — but it is the most molecularly precise hypothesis yet proposed for a microbiome-depression link.
A New Biomarker Candidate
The practical implications extend to diagnosis. DEA and the altered lipid molecule it produces could potentially serve as biomarkers to identify a subtype of depression driven by this specific inflammatory pathway.
This matters because depression is not a single disease. It is a heterogeneous syndrome with multiple biological subtypes, and the failure of many antidepressant trials may partly reflect the fact that treatments effective for one subtype are being tested in mixed populations. A biomarker that identifies the "inflammatory subtype" of depression could enable precision psychiatry — matching patients to treatments most likely to work for their specific biology.
Treatment Implications: Targeting the Immune System
The findings also raise the possibility that immune-modulating treatments could be effective for the subtype of depression driven by this pathway. Several clinical trials are already underway testing anti-inflammatory agents — including IL-6 receptor antagonists — in depression. The Harvard findings provide a mechanistic rationale for why these approaches might work in specific patient subgroups.
More speculatively, interventions targeting M. morganii abundance — through diet, probiotics, or targeted antimicrobials — or reducing DEA exposure through lifestyle and consumer product choices could represent upstream preventive strategies.
The Broader Framework: Gut Bacteria as Chemical Factories
Perhaps the most important contribution of this study is methodological. The researchers have demonstrated a framework for investigating how gut bacteria influence human physiology through the production of bioactive metabolites — chemical signals that cross the gut barrier and interact with the immune system, the nervous system, and other organs.
"Now that we know what we're looking for, I think we can start surveying other bacteria to see whether they do similar chemistry and begin to find other examples of how metabolites can affect us." — Jon Clardy
Contextualising the Evidence
It is important to note what this study does and does not establish. It identifies a plausible molecular mechanism in cell and animal models; it does not prove that DEA-M. morganii interactions cause depression in humans. The prevalence of this mechanism in the general population is unknown. DEA is ubiquitous in the environment, but the degree to which it reaches the gut at concentrations sufficient to alter M. morganii lipid chemistry in vivo requires further investigation.
Conclusion
The Harvard study represents a significant step toward a molecular understanding of how the gut microbiome influences mental health. By identifying a specific bacterium, a specific environmental contaminant, a specific altered molecule, and a specific inflammatory signal, it transforms a statistical association into a mechanistic hypothesis amenable to experimental testing and therapeutic targeting.
For the millions of people living with major depressive disorder — particularly those who have not responded to conventional antidepressants — the prospect of a gut-based diagnostic and therapeutic approach is a meaningful advance. The path from this discovery to clinical application is long, but the direction of travel is now clearer.
Sources: Harvard Medical School / Journal of the American Chemical Society, 25 April 2026 (DOI: 10.1021/jacs.4c15158); ScienceDaily, 25 April 2026; NIH Research Matters, 29 April 2026.