The search for interventions that can meaningfully slow or reverse biological aging has consumed billions of research dollars and produced countless false starts. But a study presented at Digestive Disease Week 2026 on 9 May offers something genuinely remarkable: evidence that restoring the gut microbiome to a youthful composition can reverse multiple hallmarks of aging in the liver and completely prevent the development of liver cancer in mice.
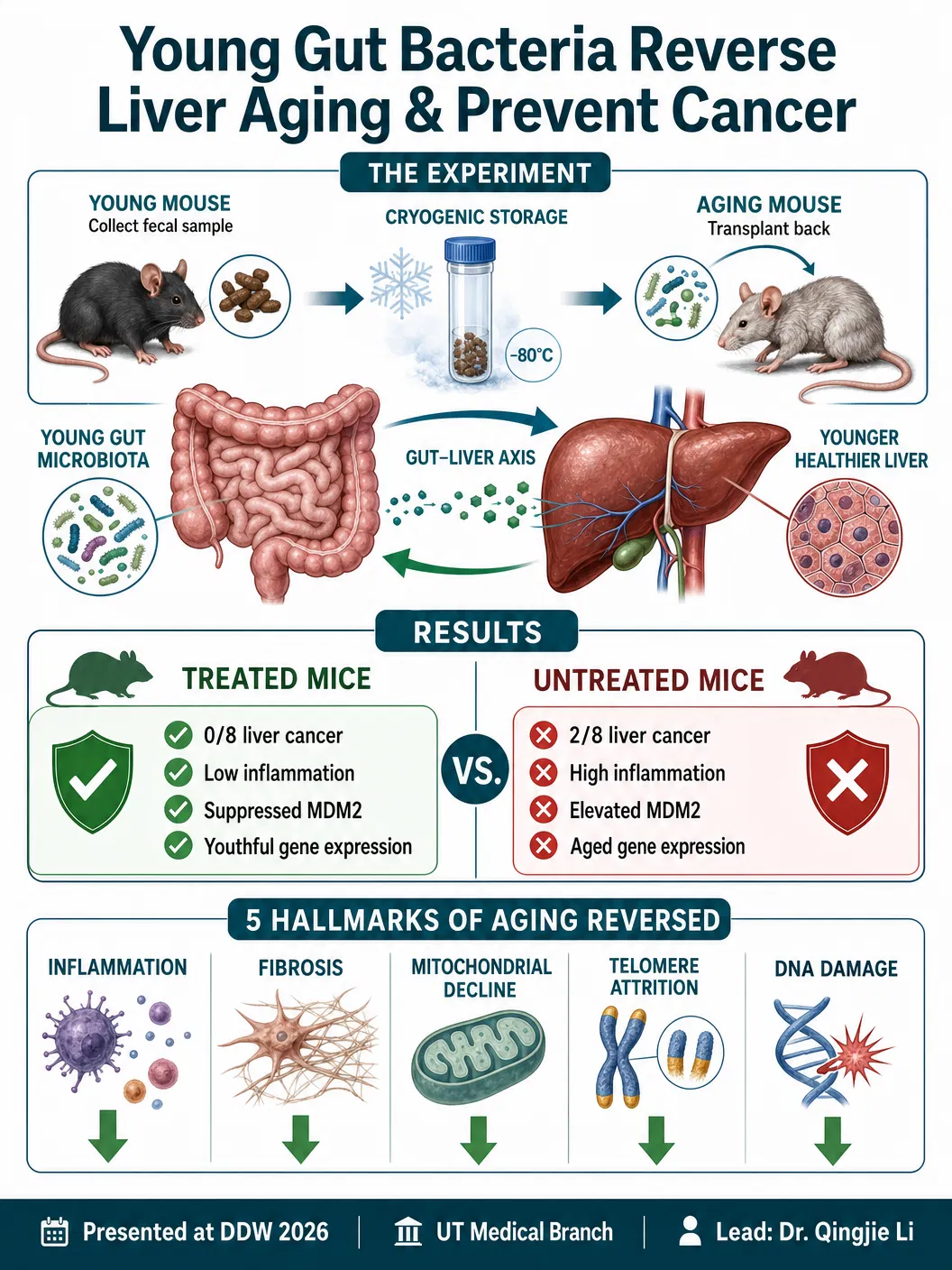
The research, led by Qingjie Li at the University of Texas Medical Branch's Division of Gastroenterology and Hepatology, employed an elegant experimental design. Scientists collected fecal samples from eight young mice and preserved them using cryogenic storage. As these same mice aged naturally over the course of the study, researchers transplanted the preserved youthful samples back into the animals through autologous fecal microbiota transplantation — meaning each mouse received its own younger microbiome rather than a donor's, eliminating the risk of immune rejection or infection.
The results were unambiguous. None of the eight mice that received their restored youthful microbiome developed liver cancer by the study's endpoint. In contrast, two of eight untreated aging mice — which received only sterilised fecal material as a control — developed liver tumours. Beyond cancer prevention, the treated mice showed dramatically lower levels of inflammation and reduced markers of liver injury compared to their untreated counterparts.
At the molecular level, the findings were equally striking. The research team examined liver tissue and discovered critical differences in MDM2, a gene already well-established in the oncology literature as a driver of liver cancer development. Young mice showed naturally low levels of MDM2 protein expression. Untreated older mice had significantly elevated MDM2 levels — consistent with their increased cancer risk. But older mice that received the microbiome restoration showed suppressed MDM2 levels that closely resembled those seen in young animals, suggesting that the gut microbiome directly influences gene expression patterns associated with cancer susceptibility.
"Restoring a more youthful microbiome can reverse several core features of aging at both the molecular and functional level," Li stated, "including inflammation, fibrosis, mitochondrial decline, telomere attrition, and DNA damage." This is not a modest claim — it encompasses virtually every major hallmark of biological aging that the geroscience community has identified over the past two decades.
The discovery emerged through scientific serendipity. Li's team was originally investigating the microbiome's effects on cardiac health in aging mice. In that earlier study, they observed that altering gut bacteria appeared to improve heart function. However, when they subsequently analysed tissue samples from multiple organs, they noticed that the liver showed even stronger beneficial effects than the heart. This unexpected observation prompted the dedicated investigation that produced the DDW 2026 findings.
The choice of autologous transplantation — using each animal's own preserved younger microbiome — represents both a scientific and a practical innovation. From a scientific perspective, it eliminates confounding variables introduced by donor microbiome variability. From a translational perspective, it suggests a potential clinical pathway: individuals could bank their own gut microbiome samples during youth or middle age, then receive them back as a therapeutic intervention later in life. This concept, sometimes called "microbiome banking," has been discussed theoretically for years but now has its strongest experimental support.
The broader implications for longevity science are substantial. The gut microbiome undergoes well-documented changes with aging — diversity decreases, certain beneficial species decline, and inflammatory species proliferate. These changes have been correlated with age-related diseases, but the directionality of the relationship has remained unclear: does the aging body cause the microbiome to deteriorate, or does the deteriorating microbiome accelerate bodily aging? Li's study provides compelling evidence for the latter interpretation. "We're learning from this work that the aging microbiome actively contributes to liver dysfunction and cancer risk rather than simply reflecting the aging process," he stated.
This finding arrives in a research landscape already primed for microbiome-based interventions. A separate study published the same week demonstrated that gut bacteria from young donors could reverse liver fibrosis markers in aged mice, while another showed that the microbiome influences brain aging through the gut-brain axis. The convergence of these findings suggests that the microbiome may represent a master regulator of systemic aging — a single intervention point that influences multiple organ systems simultaneously.
Li emphasised that the findings remain limited to animal research and cannot yet be applied to humans. However, the team is actively planning first-in-human clinical trials, which would represent the first controlled test of autologous microbiome restoration as an anti-aging intervention. If successful, such trials could establish a new therapeutic category at the intersection of gastroenterology, oncology, and geroscience.
References: Li Q, et al. Presented at Digestive Disease Week (DDW) 2026, 9 May 2026. ScienceDaily, "Scientists reversed liver aging with young gut bacteria in stunning study," 9 May 2026. DDW 2026 Conference Proceedings.