The COVID-19 pandemic exposed, with brutal clarity, the cost of unpreparedness: more than seven million confirmed deaths, trillions of dollars in economic disruption, and a global scramble to develop vaccines in real time against a pathogen that had been circulating for months before the world mobilised. The central lesson — that proactive investment in platform technologies and pre-positioned vaccine candidates saves lives and money — has since reshaped the architecture of global biosecurity. In 2026, that lesson is being applied to the threat that many virologists consider the most likely source of the next major pandemic: H5N1 highly pathogenic avian influenza.
The Scale of the Current Threat
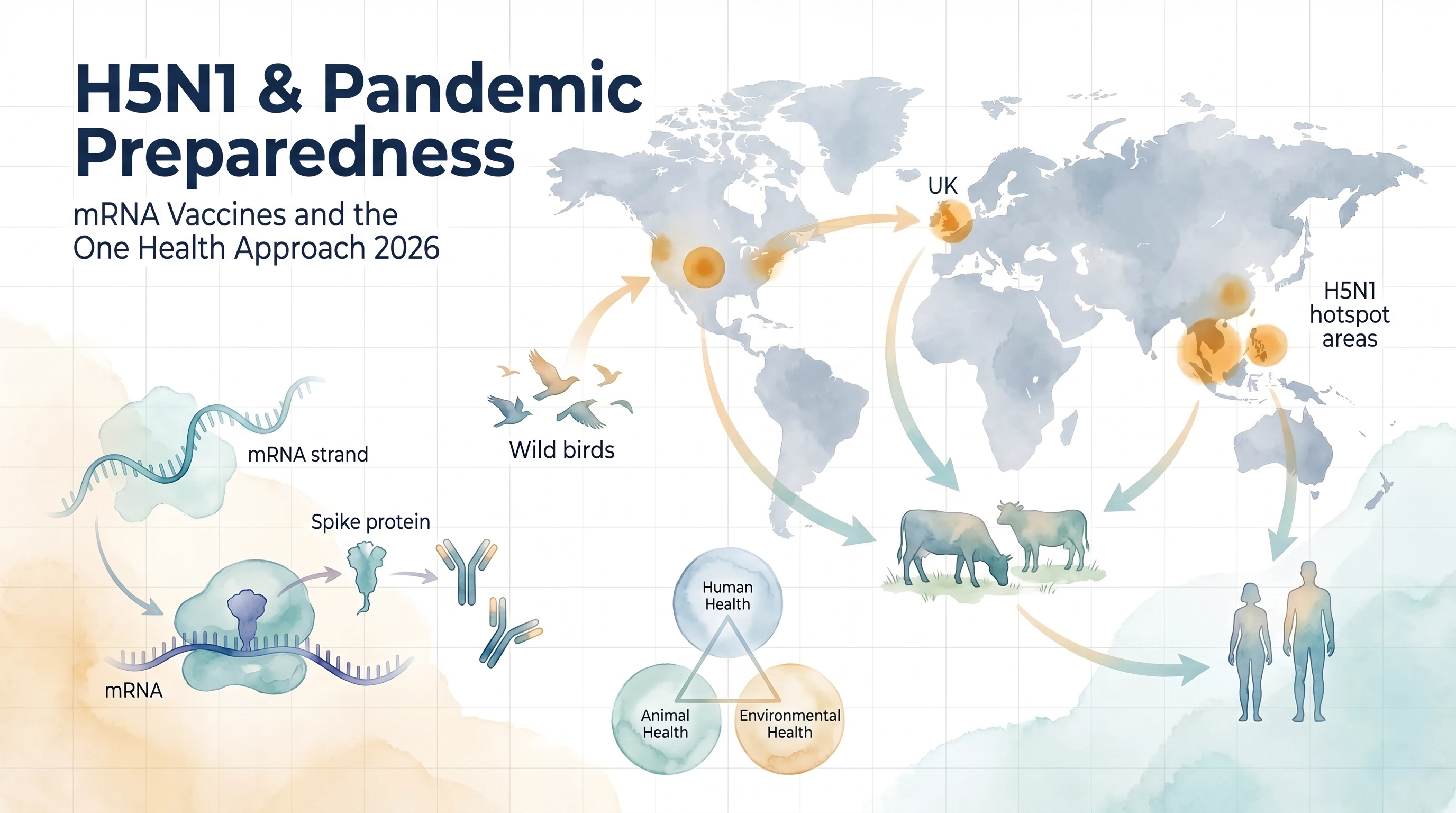
H5N1 is not a new virus. It has circulated in wild birds since the 1990s and caused periodic outbreaks in poultry flocks and sporadic human infections for decades. What has changed is its geographic reach and host range. Since 2024, the virus has been confirmed in 116 human cases globally, almost all linked to close contact with infected animals. More alarmingly, H5N1 has now been detected in dairy cattle herds across the United States, in marine mammals, and in a widening array of wild bird species — a pattern that increases the probability of the adaptive mutations required for efficient human-to-human transmission.
The biosecurity community's assessment is sober but not alarmist. The virus does not yet move easily between people. But the evolutionary pressure is real, the surveillance gaps are significant, and the consequences of being caught unprepared — as the world was in 2019 — would be catastrophic.
The mRNA Response
On 22 April 2026, CEPI (the Coalition for Epidemic Preparedness Innovations) announced the launch of the world's first pivotal Phase 3 trial of an mRNA-based H5N1 vaccine candidate. Developed by Moderna, the vaccine — mRNA-1018 — targets the A(H5N1) strain using the same messenger RNA platform that underpinned the company's highly effective COVID-19 shot. CEPI has committed up to $54.3 million to advance the candidate to licensure and to ensure equitable global access if a pandemic were to emerge.
The trial, enrolling approximately 4,000 adults across the United Kingdom and the United States, is notable not only for its scientific ambition but for its operational design. Recruitment is being conducted primarily through community health clinics rather than traditional hospital sites — a deliberate shift toward decentralised trial infrastructure that proved its value during COVID-19 and that is now being institutionalised as a preparedness norm. Moderna has also pledged that 20 percent of its H5 pandemic vaccine manufacturing capacity will be allocated for timely supply to low- and middle-income countries at affordable prices if the vaccine is licensed, addressing one of the most persistent equity failures of the COVID-19 response.
"We know that the A(H5N1) strain is evolving and spreading across animal species, and though it does not yet move easily between humans, we have to treat human-to-human transmission as a real possibility. This trial is our proactive attempt to shield against that possibility." — Dr Rebecca Clark, National Coordinating Investigator
The Broader Biosecurity Landscape
The H5N1 trial is the most visible element of a broader reconfiguration of global pandemic preparedness in 2026. The IPEA (International Pathogen Surveillance Network) consultations, whose policy brief was published on 21 April 2026, are pressing member states to strengthen genomic surveillance infrastructure and data-sharing agreements — the early-warning systems that determine whether an emerging outbreak is detected in days or months. A parallel NIH-published analysis has highlighted the dual-use implications of generative AI and protein design for biosecurity, noting that the same computational tools accelerating vaccine development can, in principle, be misused to design novel biological agents — a challenge that demands proactive governance frameworks rather than reactive regulation.
The One Health paradigm — which recognises that human, animal, and environmental health are inextricably linked — is gaining institutional traction. The H5N1 situation is a textbook illustration of its importance: the virus circulates in wild birds, amplifies in domestic poultry and cattle, and occasionally spills over into humans at the animal-human interface. Effective biosecurity requires coordinated surveillance across all three domains simultaneously, not siloed responses from separate public health, veterinary, and environmental agencies.
Africa's Particular Vulnerability
From a biosecurity perspective, the African continent faces compounded risks. Surveillance infrastructure remains uneven, cold-chain logistics for vaccine distribution are constrained, and the burden of existing infectious diseases — HIV, tuberculosis, malaria — competes for limited public health resources. The commitment by Moderna and CEPI to reserve manufacturing capacity for low- and middle-income countries is a meaningful step, but it must be accompanied by investment in the laboratory networks, trained biosecurity personnel, and regulatory frameworks that translate vaccine availability into vaccine delivery.
The architecture of pandemic preparedness being built in 2026 — mRNA platforms, decentralised trials, federated surveillance, equitable access commitments — represents the most serious global investment in biological security since the establishment of the International Health Regulations. Whether it will be sufficient depends, ultimately, on the political will to sustain that investment between crises rather than only during them.
Sources: CEPI — How a rapid-response bird flu vaccine trial is shaping global preparedness (April 22, 2026); CDC H5 Bird Flu Human Case Data (2024–2026); REACT Group — Why the IPEA matters now (April 21, 2026); NIH PMC — Protein design, generative AI and biological security (2026).