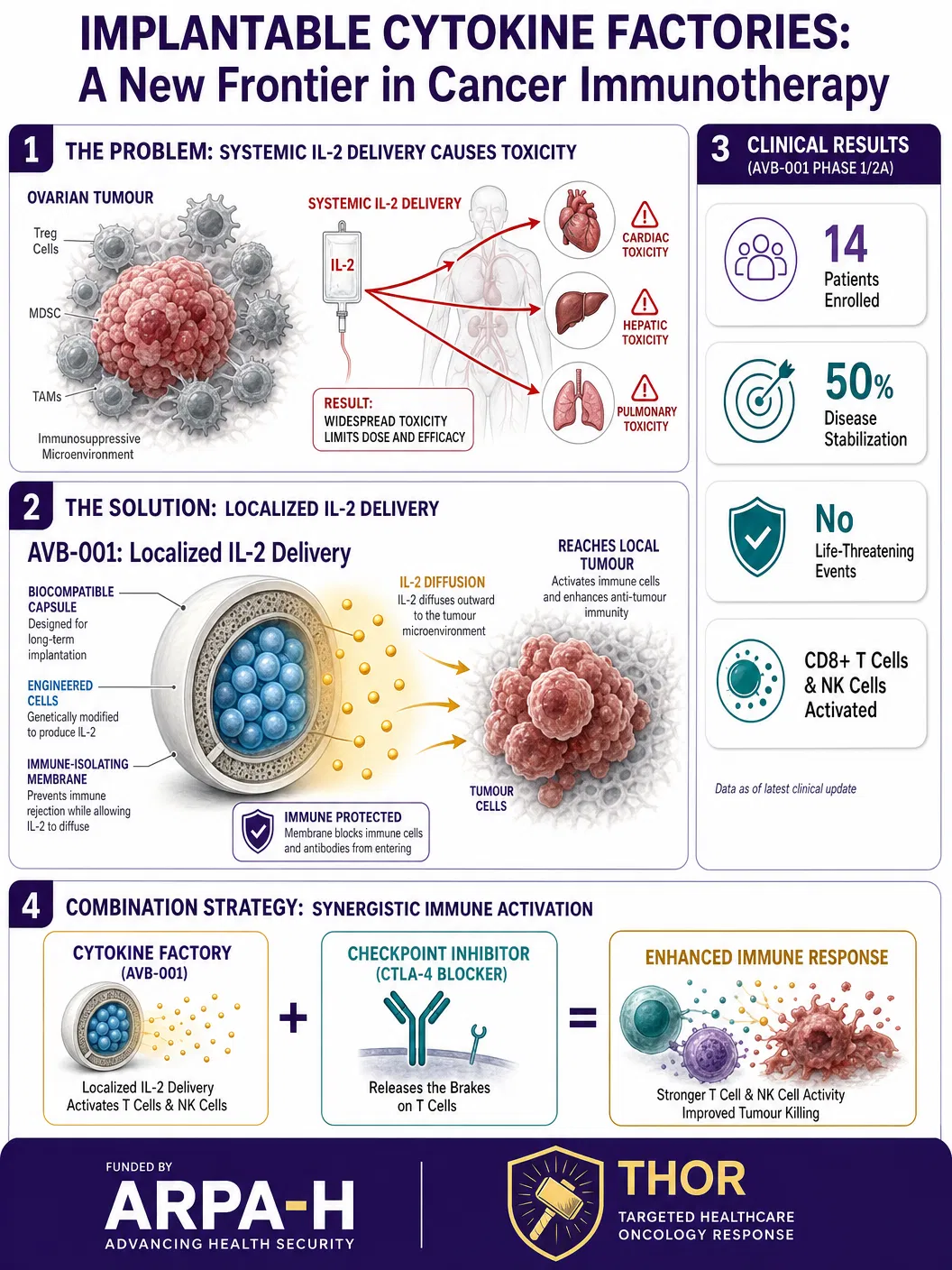
The promise of immunotherapy — harnessing the body’s own immune system to destroy cancer — has transformed outcomes in melanoma, lung cancer, and several other malignancies over the past decade. Yet for ovarian cancer, particularly the aggressive high-grade serous subtype that accounts for most deaths, immunotherapy has remained stubbornly ineffective. The tumour microenvironment in ovarian cancer is profoundly immunosuppressive, and systemic delivery of immune-activating drugs like interleukin-2 (IL-2) produces life-threatening toxicity long before therapeutic concentrations reach the tumour site.
On 12 May 2026, researchers at Rice University and The University of Texas MD Anderson Cancer Center published results in Clinical Cancer Research that may fundamentally alter this equation. Their approach bypasses the systemic delivery problem entirely: rather than flooding the entire body with immune-activating cytokines, they implant tiny capsules containing engineered cells directly into the abdominal cavity where ovarian cancer resides. These “cytokine factories” continuously produce IL-2 at the site of disease, creating a localised immune activation zone while sparing the rest of the body from toxicity.
The therapy, designated AVB-001, consists of biocompatible capsules containing genetically engineered cells programmed to secrete IL-2. The capsules are implanted via a minimally invasive laparoscopic procedure — a routine surgical technique that requires only small incisions and brief anaesthesia. Once in place, the encapsulated cells begin producing IL-2 continuously, bathing nearby tumour tissue in immune-stimulating signals for approximately one week before activity naturally declines.
The first-in-human Phase I dose-escalation trial enrolled fourteen patients with platinum-resistant high-grade serous ovarian cancer — a population with extremely limited treatment options and poor prognosis. The results were encouraging on multiple fronts. No life-threatening treatment-related adverse events were observed, and no maximum tolerated dose was reached, suggesting the approach has a wide therapeutic window. Half of the patients achieved disease stabilisation, including several with prolonged periods of clinical benefit.
“Traditional IL-2 therapy has shown potent antitumor activity, but its clinical use has been limited by severe side effects and delivery challenges,” explained Omid Veiseh, professor of bioengineering at Rice and senior author on the study. “This platform allows us to localise and sustain cytokine exposure directly where tumours reside while minimising systemic toxicity.”
The immunological data were particularly compelling. Detailed immune analyses revealed that AVB-001 successfully activated CD8+ cytotoxic T cells and natural killer (NK) cells — the immune system’s primary tumour-killing effectors — without simultaneously expanding regulatory T cells. This distinction is crucial: conventional systemic IL-2 therapy paradoxically activates both killer cells and the regulatory cells that suppress them, often negating the therapeutic benefit. The localised delivery approach appears to selectively tip the balance toward tumour destruction.
Perhaps most intriguingly, the researchers observed dose-dependent upregulation of CTLA-4, an immune checkpoint protein, on T cells exposed to the implanted factories. This finding suggests a natural combination strategy: pairing cytokine factories with checkpoint inhibitor drugs like ipilimumab could potentially amplify antitumour responses beyond what either approach achieves alone.
“What is exciting is that we are not just delivering a drug — we are programming a microenvironment,” said Dr. Amir Jazaeri, professor of gynecologic oncology at MD Anderson. “This opens the door to combination strategies that could amplify immune responses in ways that have not been feasible before.”
The research was funded by the Advanced Research Projects Agency for Health (ARPA-H) through its Targeted Hybrid Oncotherapeutic Regulation (THOR) project — a programme specifically designed to accelerate breakthrough cancer therapies from concept to clinic. The involvement of ARPA-H, modelled after DARPA’s high-risk/high-reward research philosophy, underscores the transformative potential that federal agencies see in this approach.
Future studies will evaluate higher doses, repeat administration strategies, and combination approaches with immune checkpoint inhibitors. If successful, the cytokine factory platform could extend beyond ovarian cancer to any solid tumour accessible by implantation — including peritoneal metastases from colorectal, gastric, and pancreatic cancers.
For the approximately 20,000 women diagnosed with ovarian cancer annually in the United States alone, this represents a genuinely novel therapeutic paradigm: not a new drug, but an entirely new way of delivering immune activation precisely where it is needed most.
References: Nash A, Veiseh O, Westin S, Jazaeri A, et al. First-In-Human Trial of Encapsulated Cell-Based Protein Producers for Localized IL-2 in Patients with High-Grade Serous Ovarian Carcinoma. Clinical Cancer Research. 2026. Rice University News Release, 12 May 2026. ARPA-H THOR Programme.