For more than a century, live animal experimentation has been the cornerstone of preclinical drug development. Mice, rats, rabbits, and non-human primates have served as biological proxies for the human body, helping researchers understand how candidate compounds behave before they ever reach a human volunteer. Yet the ethical, financial, and scientific limitations of this paradigm have long been recognised. Animals suffer. Experiments are expensive. And critically, animal models often fail to predict human outcomes with sufficient accuracy — a fact underscored by the sobering statistic that more than 90% of drugs that succeed in animal trials still fail in human clinical trials.
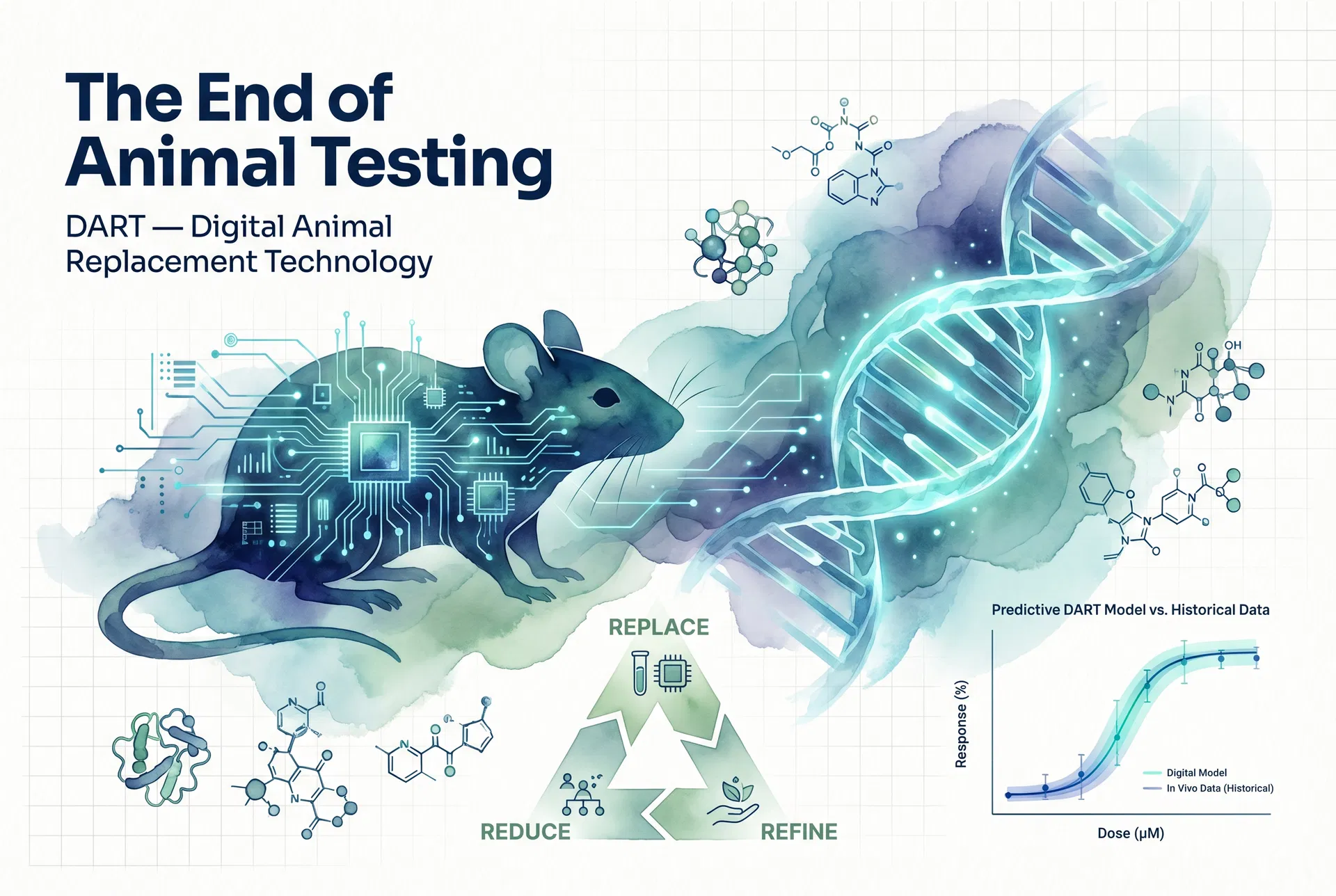
A new generation of computational biology platforms is now challenging this status quo, and among the most ambitious is Invitron — an educational simulations platform built around the principle that digital, AI-powered animal models can replace, reduce, and refine the use of live animals in research. Invitron's flagship product, InSilico Models, is a precision digital animal modelling environment that is fully compliant with the FDA Modernization Act 2.0 and aligned with the 3Rs ethical framework (Replace, Reduce, Refine) mandated by EU Directive 2010/63/EU and NC3Rs guidelines.
What Invitron Offers
At its core, Invitron provides researchers with a unified platform spanning three integrated modules. The Animal Models module offers 16 research species — from the ubiquitous C57BL/6J mouse and Sprague-Dawley rat to zebrafish, non-human primates, pigs, dogs, and cattle — each available in multiple genetically-engineered strains. Researchers can apply CRISPR-based knockout, knockin, and point mutations across 12 or more disease-relevant genes per species and observe predicted phenotypic consequences instantly, without a single animal entering a laboratory.
The ML Periodic Table module provides an interactive browser of more than 40 machine learning and deep learning algorithms, organised by category, task type, and use case. Researchers can select any algorithm to enhance DART (Digital Animal Replacement Technology) predictions, layering their own experimental data on top of the platform's pre-trained models to improve predictive accuracy for their specific compound or disease area.
The Upload & Train module completes the loop: researchers upload their own CSV or Excel experimental data, map columns to biological variables, select an ML algorithm, and train a custom model in minutes. This means that Invitron is not a static database but a living, adaptive research environment that improves with every dataset contributed.
The DART Framework
Invitron's analytical backbone is the DART (Digital Animal Replacement Technology) framework, a four-stage pipeline that mirrors the classical drug development workflow. The pipeline begins with Target Discovery, proceeds through In-Vitro Platform simulation, advances to DART Analysis (which generates nine chart types including dose-response curves, biomarker timelines, and translational scoring), and concludes with Clinical Translation — providing AI-powered human predictability scores alongside IC₅₀, EC₅₀, and LD₅₀ pharmacokinetic parameters.
The organ-on-chip component of the platform models six organs simultaneously — liver, heart, kidney, brain, lung, and gut — with real-time biomarker monitoring. For the liver, this means tracking ALT and AST; for the heart, Troponin I and BNP; for the kidney, creatinine and BUN. This multi-organ toxicity profiling is particularly valuable for identifying off-target effects that single-organ animal studies routinely miss.
Regulatory Alignment
Invitron's compliance posture is noteworthy. The FDA Modernization Act 2.0, signed into US law in December 2022, explicitly removed the requirement that drugs must be tested in animals before human trials — opening the door for validated computational alternatives. Invitron has built its platform to meet this new regulatory reality, alongside ICH S7A/S7B cardiac safety guidelines and the NC3Rs framework. For researchers in both academic and pharmaceutical industry settings, this means that data generated on Invitron can be presented to regulators as part of a credible preclinical dossier.
The platform currently supports 29 drug compounds across 8 disease areas, with pharmacokinetic parameters for compounds ranging from Imatinib (Gleevec) and Doxorubicin in oncology to Remdesivir and Oseltamivir in infectious disease, Metformin in metabolic disorders, and Adalimumab (Humira) in autoimmune conditions.
Why This Matters for Africa and the Global South
For researchers in resource-constrained settings — including much of sub-Saharan Africa — the cost and logistical complexity of maintaining live animal facilities represents a near-insurmountable barrier to preclinical research. Invitron's cloud-based, token-accessed platform removes this barrier entirely. A researcher in Nairobi, Lagos, or Kampala can now run a fully compliant DART analysis on a novel antimalarial compound using a validated murine model, generate publication-quality pharmacokinetic data, and export results in CSV, JSON, or report format — all without a single animal or a single piece of laboratory equipment.
This democratisation of preclinical research infrastructure is, arguably, Invitron's most transformative contribution to global science. Explore the platform at https://www.invitron.org.