A Single Infusion That Rewrites the Rules
On 27 April 2026, Intellia Therapeutics announced results that will be cited in medical textbooks for decades: a single intravenous dose of lonvo-z (lonvoguran ziclumeran) reduced hereditary angioedema (HAE) attacks by 87% compared to placebo in the global Phase 3 HAELO trial. More striking still, 62% of treated patients were entirely attack-free for the six-month evaluation window, versus just 11% on placebo.
This was not merely a clinical win. It was the first Phase 3 data ever reported for an in vivo gene-editing therapy — a therapy that edits a patient's DNA directly inside their living body, without removing cells, modifying them in a laboratory, and returning them. The distinction matters enormously.
What Is Hereditary Angioedema?
HAE is a rare inherited disorder caused by mutations that lead to excessive production of bradykinin, a peptide that makes blood vessels abnormally leaky. The result is unpredictable, potentially life-threatening swelling attacks affecting the abdomen, limbs, face, and — most dangerously — the upper airway. Laryngeal attacks can be fatal if untreated.
Current treatments require lifelong dosing: daily oral tablets, regular subcutaneous injections, or intravenous infusions every few weeks. They work, but they are burdensome, expensive, and breakthrough attacks still occur. Patients live under the constant shadow of the next episode.
How Lonvo-z Works: Editing the Source
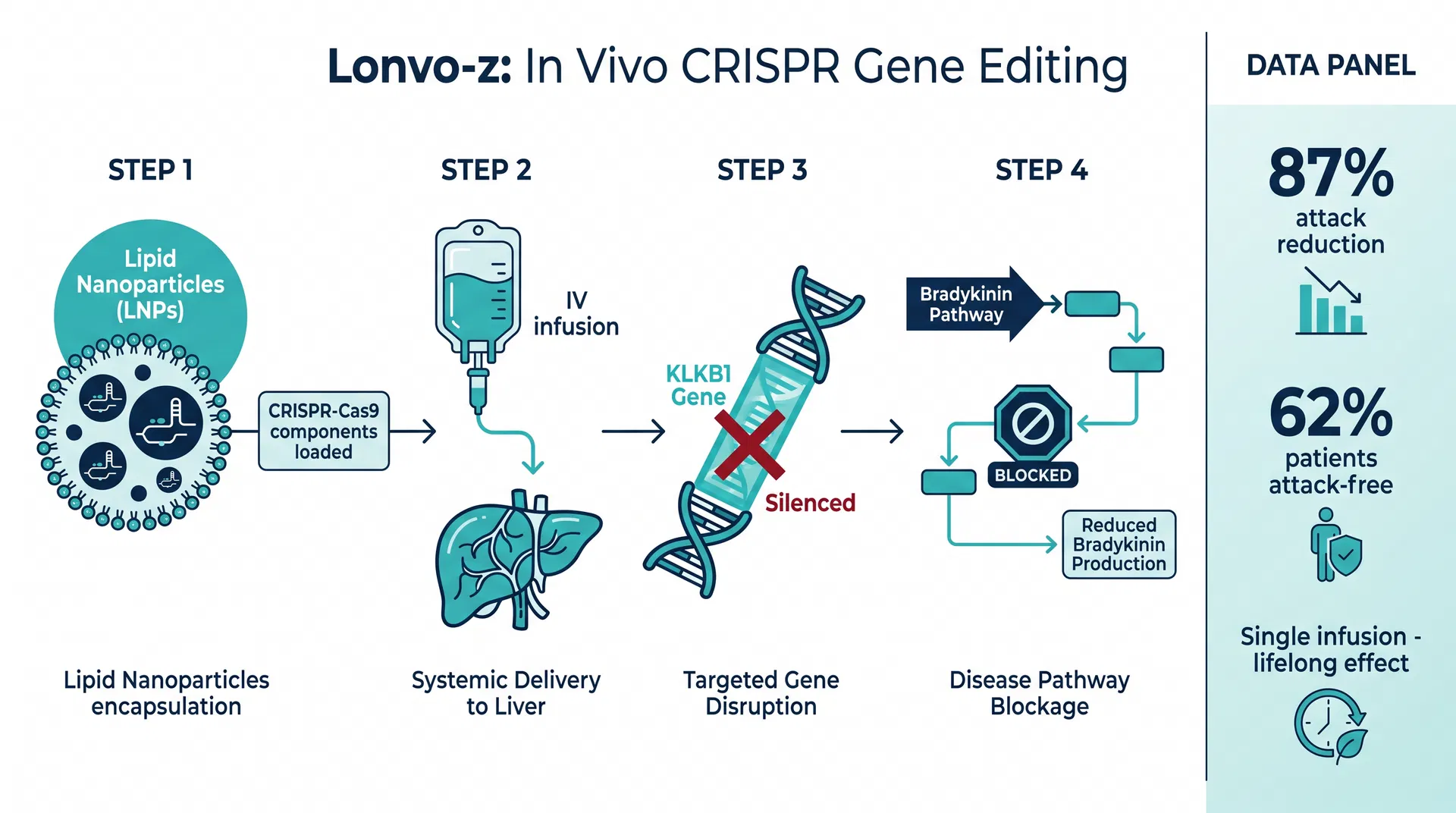
Lonvo-z takes a fundamentally different approach. Rather than blocking the bradykinin pathway repeatedly with chronic medication, it aims to permanently silence the gene that drives the problem.
The therapy targets KLKB1, the gene encoding plasma prekallikrein — the precursor of kallikrein, the enzyme that generates bradykinin. Using lipid nanoparticles (the same delivery technology that made mRNA COVID-19 vaccines possible) to carry CRISPR-Cas9 components into liver cells, lonvo-z introduces a precise frameshift edit in KLKB1. This disabling edit permanently reduces prekallikrein production after a single outpatient infusion.
The mechanism is elegant in its simplicity: remove the upstream source of the problem, and the downstream cascade of swelling attacks cannot occur.
The HAELO Trial: By the Numbers
The Phase 3 HAELO trial enrolled 80 adults and adolescents (≥16 years) with Type I or Type II HAE. Participants were randomised to a single 50 mg intravenous infusion of lonvo-z (n=52) or placebo (n=28). Key results:
| Endpoint | Lonvo-z | Placebo |
|---|---|---|
| Mean monthly attack rate (weeks 5–28) | 0.26 | 2.10 |
| Attack rate reduction vs placebo | 87% | — |
| Patients entirely attack-free | 62% | 11% |
| Patients free of long-term prophylaxis at data cut | 100% | — |
All key secondary endpoints were met. Treatment-emergent adverse events were predominantly mild-to-moderate infusion-related reactions, headache, and fatigue. No serious adverse events occurred in the lonvo-z arm.
Long-term Phase 1/2 data presented at the American Academy of Allergy, Asthma & Immunology meeting in February 2026 reinforce durability: in 32 patients followed for a median of 12.2 months (range 2.4 months to 3 years), mean reductions in plasma kallikrein of 86–96% were sustained throughout follow-up.
The Regulatory Fast Lane
Lonvo-z carries a formidable regulatory portfolio:
- FDA Regenerative Medicine Advanced Therapy (RMAT) designation — permits rolling submission and expedited review
- Orphan Drug Designation in the US and EU
- UK MHRA Innovation Passport
- EMA PRIME designation
Intellia has already initiated a rolling BLA submission and expects to complete it in the second half of 2026, with a potential US launch in the first half of 2027.
Why This Matters Beyond HAE
The significance of lonvo-z extends far beyond the approximately 1 in 50,000 people affected by HAE. It is a proof of concept for in vivo CRISPR medicine at the highest level of clinical evidence.
The first approved CRISPR therapy, Vertex's Casgevy for sickle cell disease, works by editing cells outside the body — a complex, expensive process requiring specialised centres. Lonvo-z demonstrates that CRISPR can work inside the body, delivered by a lipid nanoparticle, in an outpatient setting, with a safety profile comparable to a standard infusion.
This opens the door to in vivo CRISPR therapies for a wide range of conditions where the target tissue is accessible to LNP delivery — the liver in particular, but potentially the lung, muscle, and central nervous system as delivery technologies mature.
"As the first Phase 3 data reported for an in vivo gene editing therapy, today's HAELO results represent a profound milestone for Intellia, the broader CRISPR and precision medicine fields and, most importantly, the HAE community." — John Leonard, President & CEO, Intellia Therapeutics
A Biosecurity and Biosafety Perspective
From a biosecurity standpoint, the lonvo-z milestone raises important dual-use considerations. The same lipid nanoparticle delivery platforms and CRISPR editing machinery that can silence a disease-causing gene could, in principle, be directed at other genomic targets. As in vivo gene editing moves from rare disease to broader application, governance frameworks will need to keep pace with the technology.
The biosafety record of lonvo-z to date is reassuring: off-target editing analyses have not identified clinically significant unintended edits, and the liver-tropism of LNPs limits systemic exposure. But long-term surveillance will be essential as the first cohorts of in vivo CRISPR patients are followed over decades.
Conclusion
Lonvo-z is not just a new drug. It is the opening chapter of a new era in medicine — one in which a single infusion can permanently rewrite the genetic instructions driving a disease. For the HAE community, it offers the prospect of a life free from the unpredictable terror of swelling attacks. For the broader field of precision medicine, it is the clearest signal yet that in vivo gene editing has arrived.
Sources: Intellia Therapeutics press release, 27 April 2026; CRISPR Medicine News, 29 April 2026; STAT News, 27 April 2026.