On 28 April 2026, three scientists received the most prestigious honour in life sciences research: the Breakthrough Prize, widely known as the "Oscars of Science." Jean Bennett, Albert Maguire, and Katherine High — all affiliated with the University of Pennsylvania and the Children's Hospital of Philadelphia (CHOP) — were awarded the $3 million prize for their foundational work in creating Luxturna, the world's first FDA-approved gene therapy for an inherited disease.
Their achievement did not happen overnight. It is the product of more than three decades of scientific persistence, clinical courage, and a willingness to pursue a field that the broader research community had largely abandoned.
The Disease: Leber Congenital Amaurosis
Luxturna treats Leber Congenital Amaurosis (LCA), a rare inherited retinal dystrophy caused by mutations in the RPE65 gene. The RPE65 protein is essential for the visual cycle — the biochemical process by which photoreceptor cells regenerate the light-sensitive pigment needed to detect light. Without functional RPE65, photoreceptors gradually degenerate, and patients experience severe vision loss from birth, progressing to complete blindness.
LCA affects approximately 2–3 per 100,000 live births. Before Luxturna, there was no treatment. Children with the condition navigated a world of permanent darkness, with parents and clinicians powerless to intervene.
The Science: Delivering a Healthy Gene Directly to the Retina
The therapy works by delivering a functional copy of the RPE65 gene directly to the retinal cells that need it. The delivery vehicle is an adeno-associated virus serotype 2 (AAV2) — a naturally occurring virus that has been engineered to carry therapeutic DNA without causing disease.
The procedure involves a sub-retinal injection: a surgeon makes a small incision in the eye and injects the AAV2 vector into the space beneath the retina, where it infects retinal pigment epithelium cells and delivers the healthy RPE65 gene. Once inside the cell, the gene is expressed, functional RPE65 protein is produced, and the visual cycle is restored.
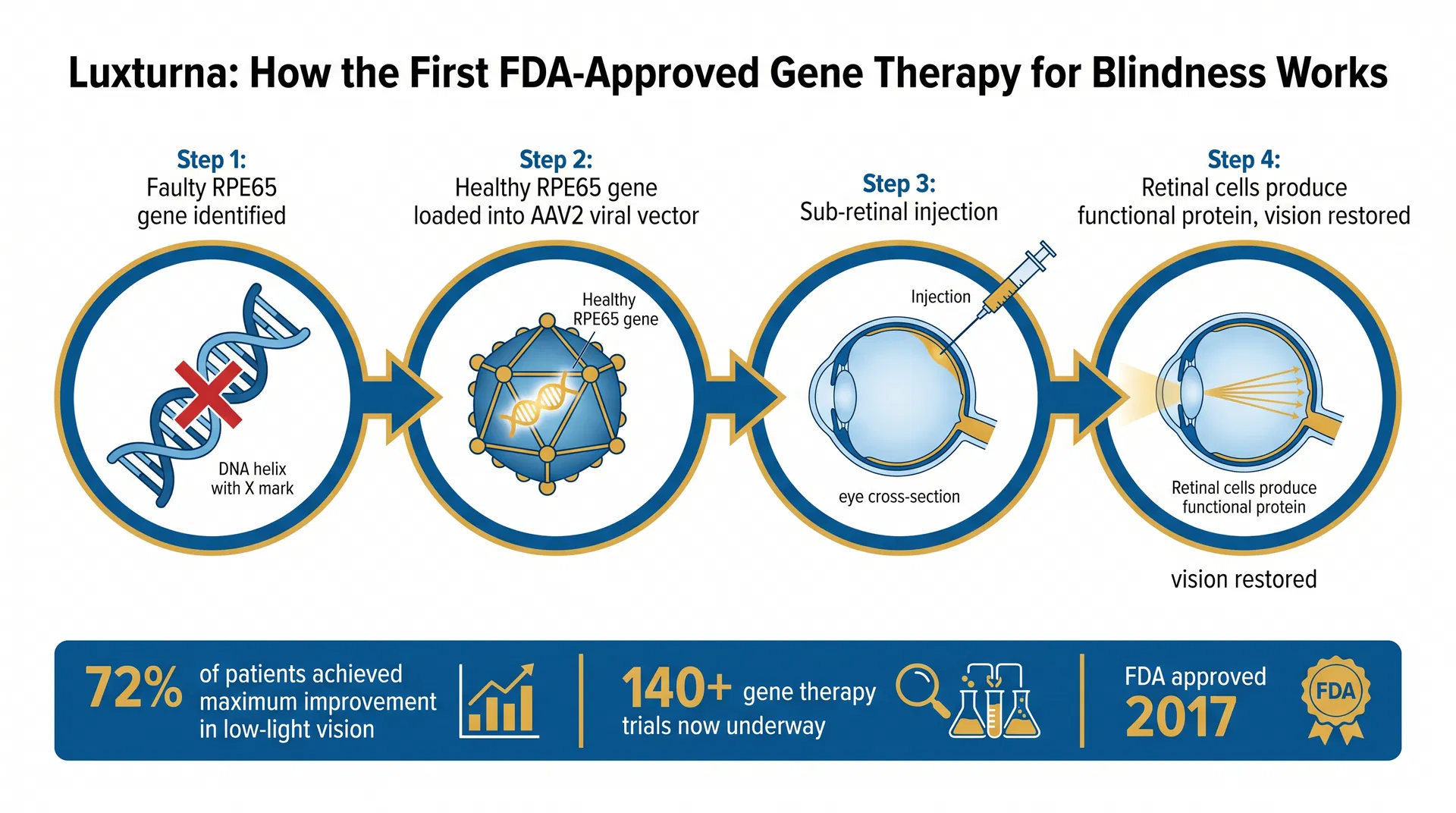
Figure 1: The four-step Luxturna process: faulty RPE65 gene identified → healthy gene loaded into AAV2 vector → sub-retinal injection → retinal cells produce functional protein and vision is restored.
| Metric | Result |
|---|---|
| Patients achieving maximum low-light vision improvement | 72% |
| FDA approval year | 2017 |
| Gene therapy trials for retinal conditions now underway | 140+ |
| Additional trials currently recruiting | 80+ |
| Diseases potentially addressable by retinal gene therapy | Macular degeneration, diabetic retinopathy, and more |
Table 1: Key outcomes and impact of the Luxturna programme.
The Moment of Discovery
The pivotal early experiments were conducted in dogs. Gustavo Aguirre at the University of Pennsylvania School of Veterinary Medicine had identified a spontaneous canine model of LCA-like blindness, which proved invaluable. Bennett and Maguire injected the experimental therapy into a single eye of each of three dogs.
The phone call that followed has become part of scientific legend. A laboratory technician called Bennett in a state of excitement: "They can see!" The dogs, which had previously bumped and tripped through an obstacle course, were now navigating it with ease — using the treated eye to guide them.
"Working without the benefit of the guidelines and precedents we now have today, we sought to solve each day's problems so that the programme would have a tomorrow. It was a bold and uncertain investment of time, effort, and resources." — Katherine High, MD
The Human Trials
The first human trial participant — a 26-year-old woman — received the sub-retinal injection at CHOP in 2007, administered by Maguire. Her twin, who had the same condition, received the treatment shortly after.
When the team assessed the 37 eligible participants from the original clinical trials, the results were transformative. Seventy-two percent reported the maximum possible improvement in a standardised low-light vision test. One patient, who had previously only been able to detect changes in light intensity, was suddenly able to navigate Philadelphia streets at night without assistance and could make out the clock on City Hall. Another patient saw a star for the first time in her life, just six days after the procedure.
The Regulatory Milestone and Its Legacy
In 2017, the FDA approved Luxturna — manufactured by Spark Therapeutics, a CHOP spinout — making it the first FDA approval of a gene therapy for an inherited disease in history. The approval opened a new regulatory pathway and demonstrated to the entire biomedical community that gene therapy was not merely theoretical but clinically viable.
The ripple effects have been enormous. More than 140 gene therapy trials for retinal conditions are now underway, targeting diseases including age-related macular degeneration and diabetic retinopathy — conditions that collectively affect approximately 30 million people in the United States alone.
"Developing cell and gene therapies has long been a top priority for our organisation," said Madeline Bell, CHOP's CEO. "This breakthrough is the result of decades of investment and collaboration, and reflects our commitment to translating scientific discoveries into therapies that will transform patients' lives."
The Breakthrough Prize and What It Represents
The Breakthrough Prize, created in 2012 by Sergey Brin, Priscilla Chan, Mark Zuckerberg, Yuri and Julia Milner, and Anne Wojcicki, is awarded annually in Life Sciences, Fundamental Physics, and Mathematics. At $3 million per award, it is the world's largest science prize. The 2026 ceremony, held in Hollywood, was co-hosted by Academy Award-winning actors Octavia Spencer and Sean Penn.
For Bennett, Maguire, and High, the prize is recognition not just of a single therapy but of a philosophy: that inherited diseases, once considered untreatable by definition, are now within the reach of molecular medicine. Their work has established the template — the vector, the delivery method, the regulatory framework, the clinical trial design — that every subsequent retinal gene therapy programme has built upon.
Source: University of Pennsylvania Almanac, April 28, 2026. The 2026 Breakthrough Prize in Life Sciences was awarded at a ceremony in Hollywood, California.