The history of engineering is, in one sense, the history of programmability. The invention of the programmable loom by Joseph-Marie Jacquard in 1804 — in which punch cards encoded the pattern of a woven fabric — was not merely a labour-saving device but a conceptual breakthrough: the realisation that complex physical processes could be controlled by abstract symbolic instructions. Charles Babbage recognised the generality of this principle and conceived the Analytical Engine. Ada Lovelace understood that the same principle could be applied to any process that could be formalised. Alan Turing proved that a universal programmable machine was theoretically possible. And the rest, as they say, is the history of computing.
Biology is undergoing an analogous transformation. The recognition that DNA is, in a precise sense, a program — a sequence of symbolic instructions that directs the construction and operation of living systems — has been the foundational insight of molecular biology since Watson and Crick. But for most of the history of molecular biology, this program was readable but not writable: scientists could sequence and interpret genetic information, but their ability to rewrite it with precision and predictability was severely limited. The development of CRISPR-Cas9 genome editing, synthetic gene circuits, and programmable transcription factors has changed this. And AI is now transforming the programmability of biology from a laboratory capability into an engineering discipline.
The Grammar of Biological Programs
To program a living system, one must first understand its language. The language of biology is not a simple linear code but a complex, context-dependent grammar in which the meaning of any genetic element depends on its sequence, its position in the genome, the regulatory environment of the cell, and the evolutionary history of the organism. Deciphering this grammar — understanding the rules by which genetic sequences are interpreted to produce biological functions — is the central challenge of systems biology.
AI is making unprecedented progress on this challenge. Deep learning models trained on large-scale functional genomics datasets are learning the grammar of gene regulation with a precision that exceeds classical bioinformatics approaches. Models such as Enformer and Borzoi can predict the transcriptional output of any DNA sequence in any cellular context, effectively learning the rules by which the genome is read. Protein language models can predict the functional consequences of any amino acid sequence, learning the grammar of protein structure and function from the statistical regularities of the evolutionary record.
These models are not just analytical tools — they are the foundation of a new kind of biological programming. By learning the grammar of biological systems, they enable the design of new genetic elements — promoters, enhancers, coding sequences, regulatory RNAs — that produce specified biological outputs with a reliability that was previously unachievable. This is the beginning of a genuinely programmable biology.
Gene Circuits: The Logic Gates of Living Systems
The most direct analogy between biological programming and electronic programming is found in the design of synthetic gene circuits — networks of genetic elements that implement logical operations in living cells. Just as electronic circuits implement Boolean logic through the combination of transistors, synthetic gene circuits implement biological logic through the combination of transcription factors, promoters, and regulatory RNAs.
The design of gene circuits that function reliably in living cells is extraordinarily challenging. Biological systems are noisy, context-dependent, and subject to evolutionary pressure that tends to degrade engineered functions over time. The design space of possible circuit architectures is astronomically large, and the relationship between circuit design and circuit behaviour is highly non-linear. These are precisely the kinds of challenges that AI is well-suited to address.
Reinforcement learning algorithms are being used to design gene circuit architectures that achieve specified input-output relationships while minimising metabolic burden and maximising robustness to noise. Generative models are being used to design novel regulatory elements — promoters, ribosome binding sites, terminators — with specified activity levels and context-independence. Bayesian optimisation algorithms are being used to tune the parameters of gene circuits in living cells, converging on optimal configurations through iterative experimental rounds.
| Circuit Type | Biological Implementation | AI Design Approach | Application |
|---|---|---|---|
| Toggle switch | Mutual repressor network | Reinforcement learning for parameter optimisation | Cell fate control, biosensors |
| Oscillator | Repressilator, activator-repressor | Evolutionary algorithms for topology search | Circadian rhythm engineering, drug delivery |
| Logic gates (AND, OR, NOT) | Transcription factor combinations | Deep learning for promoter design | Biosensors, therapeutic cell programming |
| Memory elements | Epigenetic switches, recombinase circuits | Bayesian optimisation | Cell lineage tracking, recording systems |
| Feedback controllers | Integral feedback circuits | Control theory + ML | Robust metabolic flux control |
| Classifiers | Multi-input transcription factor networks | ML-guided combinatorial design | Cancer cell detection, therapeutic targeting |
Cell Programming: Therapeutic Applications
The most immediately consequential application of programmable biology is in medicine — specifically in the engineering of cells that can detect disease states and respond with therapeutic interventions. CAR-T cell therapy, in which patient T cells are reprogrammed to recognise and kill cancer cells, is the most clinically advanced example of this approach, but it represents only the beginning of what programmable cellular medicine can achieve.
AI is accelerating the development of next-generation cell therapies by enabling the design of more sophisticated genetic programs that give therapeutic cells greater precision, persistence, and safety. Multi-input logic circuits can program therapeutic cells to activate only in the presence of multiple disease-specific signals, reducing off-target toxicity. Feedback control circuits can regulate the activity of therapeutic cells in response to real-time signals from the disease environment. Memory circuits can enable therapeutic cells to record their history of antigen encounters, informing their future behaviour.
Beyond oncology, programmable cell therapies are being developed for autoimmune diseases, metabolic disorders, and infectious diseases. In each case, AI is providing the design intelligence needed to navigate the vast space of possible genetic programs and identify those most likely to achieve the desired therapeutic outcome.
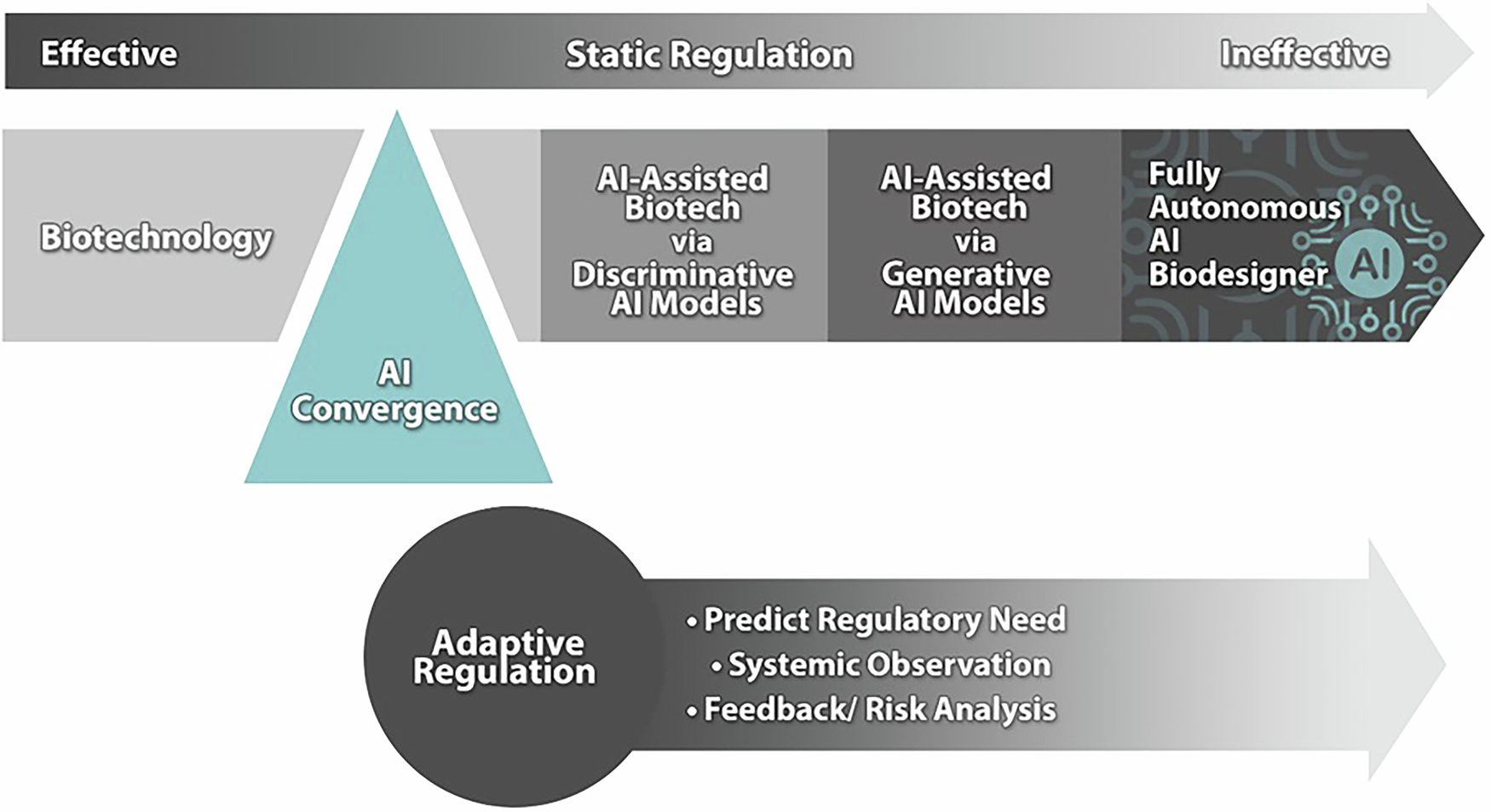
Governance of Programmable Biology
The programmability of biology raises governance challenges that are qualitatively different from those posed by earlier biotechnologies. When biological systems can be designed to specification — when the gap between intention and outcome is narrowed by AI-powered design tools — the range of possible applications, both beneficial and harmful, expands dramatically. The same tools that enable the design of therapeutic gene circuits could, in principle, be used to design pathogenic ones. The same AI systems that optimise metabolic pathways for bioplastic production could be used to optimise the production of toxic compounds.
Governance frameworks for programmable biology must therefore be designed with this dual-use potential in mind. They must be capable of assessing not just the properties of specific biological products but the capabilities of the design tools and platforms that produce them. They must be adaptive, capable of keeping pace with the rapid evolution of AI-powered biological design. And they must be international, because the programmability of biology is a global phenomenon that cannot be governed effectively by any single nation or institution.
The vision of programmable biology — of living systems that can be designed, modified, and controlled with the precision of engineered systems — is both exhilarating and sobering. It represents the fulfilment of a promise that synthetic biology has been making for two decades. And it demands, in equal measure, the development of governance frameworks worthy of the responsibility it confers.