Fungal infections represent one of medicine's most underappreciated threats. Aspergillus fumigatus, a ubiquitous airborne mould whose spores every human inhales daily, kills tens of thousands of immunocompromised patients worldwide each year — and existing antifungal drugs are increasingly failing due to resistance. A study published in Science Advances on 8 May 2026 reveals a discovery that could fundamentally change how we approach this problem: a previously unknown protein "switch" inside immune cells that determines whether the body successfully destroys fungal invaders or allows them to survive despite mounting a seemingly aggressive immune response.
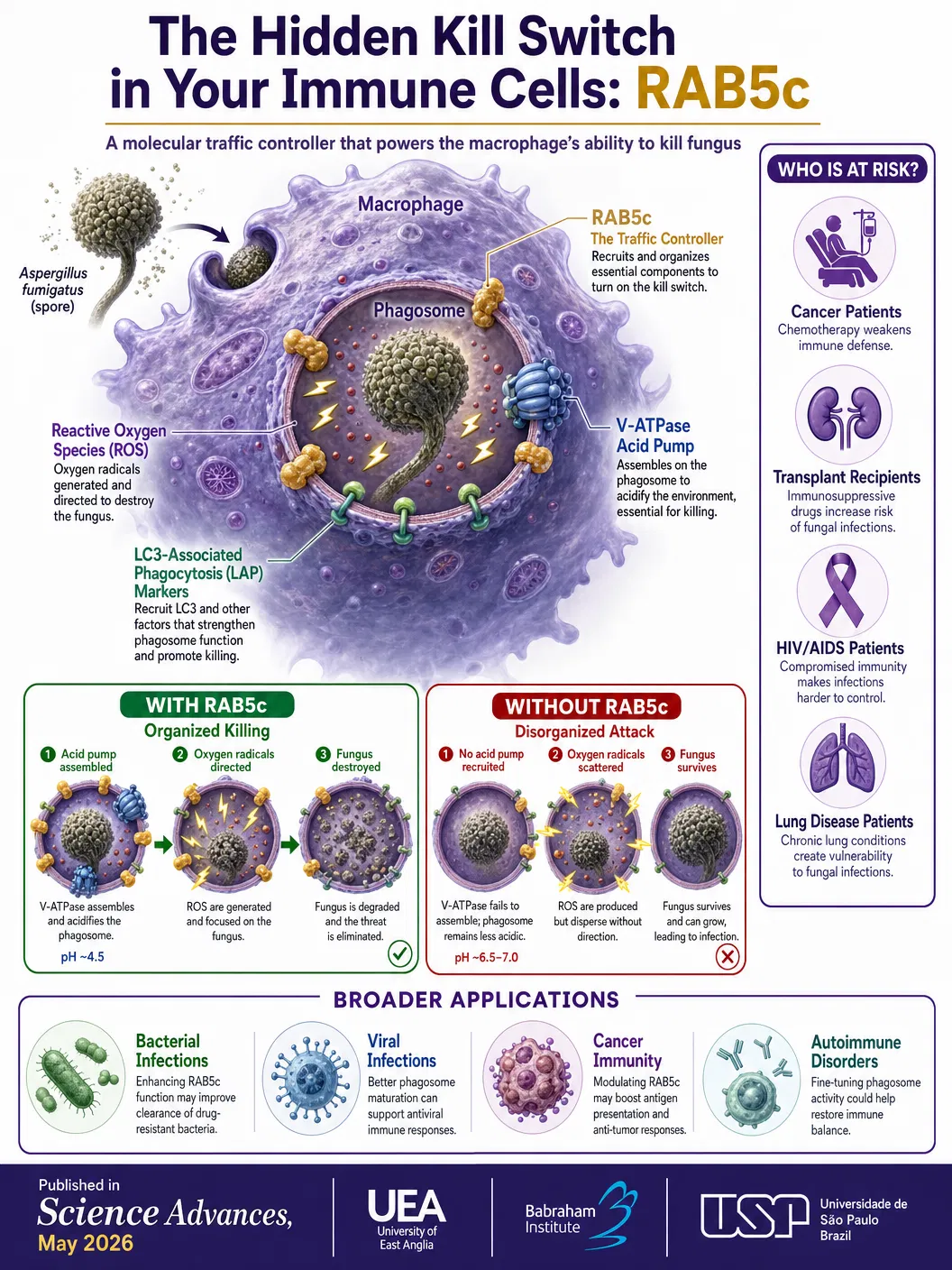
The research, conducted by scientists at the University of East Anglia, the Babraham Institute, and the Universidade de São Paulo, identifies RAB5c as the critical orchestrator of fungal killing within macrophages — the immune system's frontline "Pac-Man" cells responsible for engulfing and destroying pathogens. The team, led by Professor Tom Wileman of UEA's Norwich Medical School, discovered that RAB5c functions as a molecular "traffic controller," ensuring that lethal enzymes and toxic molecules are delivered to the correct intracellular compartment at precisely the right moment to destroy engulfed fungal spores.
The mechanism works through a process called LC3-associated phagocytosis, or LAP. When a macrophage swallows a fungal spore, it forms a sealed internal compartment called a phagosome. Under normal conditions, this compartment acidifies, fills with toxic reactive oxygen species, and deploys the LAP machinery to destroy the trapped pathogen. RAB5c, the researchers found, is essential for assembling a microscopic "acid pump" called V-ATPase on the phagosome membrane. This pump creates the acidic environment necessary for reactive oxygen species to trigger the final killing cascade.
The study's most surprising finding was what happens when RAB5c is absent. Rather than simply failing to mount an immune response, macrophages lacking RAB5c actually produced more toxic oxygen molecules than normal — yet the fungus survived. "This is because those toxic molecules were not properly harnessed," Wileman explained. "RAB5c plays a key role in assembling the V-ATPase acid pump, which allows oxygen radicals to trigger the final killing steps. Without the pump, the chemicals rage uselessly while the fungus remains alive."
This paradox — more firepower but less killing — has profound implications for understanding why some patients' immune systems fail despite appearing to mount vigorous responses. It suggests that the problem in many cases of invasive aspergillosis is not insufficient immune activation but rather disorganised immune execution. The immune system is firing all its weapons, but without RAB5c coordinating the attack, those weapons miss their target.
To validate their laboratory findings, the team infected mice with Aspergillus fumigatus. Animals unable to use the LAP-RAB5c pathway had far higher fungal loads in their lungs, more tissue damage, and stronger inflammatory responses — a pattern that mirrors the clinical presentation of invasive aspergillosis in immunocompromised humans. In contrast, mice with fully functioning RAB5c-dependent pathways cleared the infection efficiently with minimal collateral damage.
The therapeutic implications are significant. Current approaches to invasive fungal infections rely primarily on antifungal drugs — azoles, echinocandins, and amphotericin B — that directly attack the fungus. However, resistance to these drugs is rising globally, and they carry substantial toxicity. The RAB5c discovery suggests an entirely different therapeutic strategy: rather than attacking the fungus directly, future treatments could enhance the patient's own immune machinery, fine-tuning it to work more efficiently against fungal invaders.
The broader relevance extends well beyond mycology. The same LAP-RAB5c pathway is involved in killing intracellular bacteria and viruses, controlling inflammation, mediating cancer immunity, and regulating autoimmune responses. Understanding how to modulate this pathway could therefore have applications across infectious disease, oncology, and autoimmune medicine. A single molecular target that influences such diverse immune functions represents an unusually promising therapeutic opportunity.
For the estimated 6.5 million people worldwide who develop serious fungal infections annually — including cancer patients undergoing chemotherapy, organ transplant recipients on immunosuppressive drugs, and individuals with HIV/AIDS — this discovery offers hope that a new generation of immune-enhancing therapies could complement or eventually replace conventional antifungals. The challenge now is translating the RAB5c mechanism into drugs that can safely boost this specific pathway without triggering excessive inflammation or autoimmune reactions.
References: Wileman T, et al. RAB5c orchestrates LC3-associated phagocytosis to promote microbicidal function of macrophages. Science Advances. 8 May 2026. EurekAlert/AAAS News Release, 11 May 2026. University of East Anglia Press Release, May 2026.