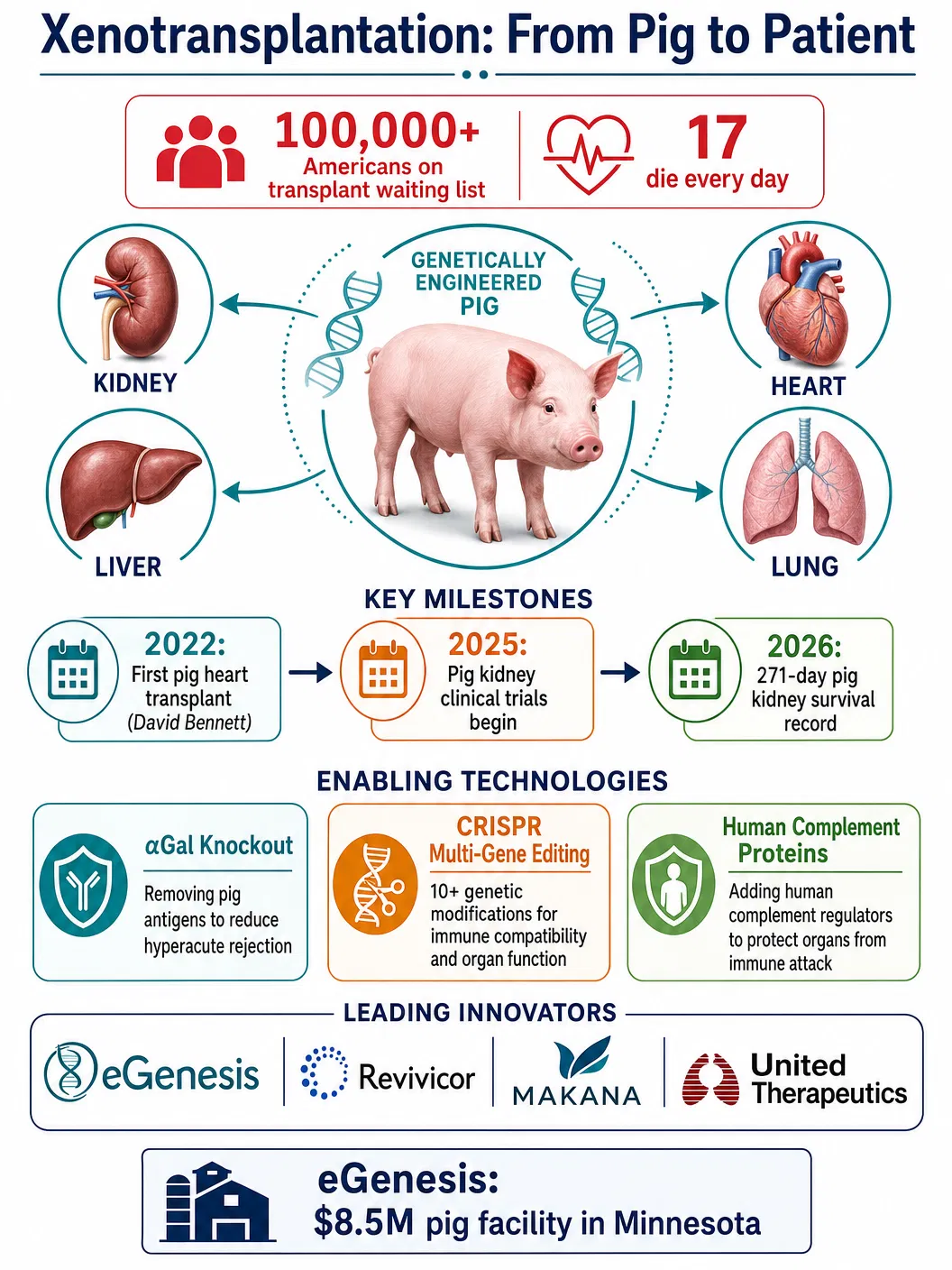
Every day in the United States, seventeen people die waiting for an organ transplant. More than 100,000 Americans are currently on the national transplant waiting list, and the gap between organ supply and demand has widened relentlessly for decades despite public awareness campaigns, policy reforms, and the expansion of living donor programmes. The fundamental arithmetic is unforgiving: there are simply not enough human organs to save everyone who needs one. But in 2026, a field that was once dismissed as science fiction is delivering results that suggest the organ shortage may finally have a solution — and it comes from pigs.
Xenotransplantation — the transplantation of organs, tissues, or cells from one species to another — has entered a period of unprecedented clinical momentum. In May 2026, USA Today reported that a man had survived 271 days with a functioning pig kidney, the longest such survival on record. In April, eGenesis, one of the leading xenotransplantation companies, paid $8.5 million for a 200-acre facility near Stewartville, Minnesota, to house genetically engineered pigs specifically bred as organ donors. And on 8 May 2026, Frontiers in Transplantation published an extensive interview with Dr. David K. C. Cooper, widely regarded as the father of modern xenotransplantation, in which he reflected on the field’s journey from speculative experimentation to clinical reality.
The scientific foundation of modern xenotransplantation rests on a single transformative technology: genetic engineering. When researchers first attempted to transplant pig organs into primates in the twentieth century, the organs were destroyed within minutes by a process called hyperacute rejection. The human immune system recognised carbohydrate molecules on pig cell surfaces — particularly alpha-galactose (αGal) — as foreign invaders and mounted an overwhelming attack involving pre-existing antibodies and the complement cascade. The organs turned black and ceased functioning almost immediately.
The breakthrough came with the development of αGal-knockout pigs — animals genetically engineered to lack the αGal carbohydrate that triggers hyperacute rejection. This single modification extended graft survival from minutes to days and then weeks. Subsequent generations of engineered pigs incorporated additional modifications: the expression of human complement-regulatory proteins to dampen immune attacks, the deletion of other pig carbohydrate antigens (Neu5Gc and SDa), and the insertion of human genes that regulate coagulation and inflammation.
Today’s most advanced donor pigs carry ten or more genetic modifications simultaneously, made possible by CRISPR-Cas9 technology, which allows researchers to edit multiple genes in a single generation rather than through years of selective breeding. Companies like eGenesis, Revivicor (a subsidiary of United Therapeutics), and Makana Therapeutics are each pursuing slightly different genetic engineering strategies, creating a competitive landscape that is accelerating progress.
The clinical milestones have been remarkable. In January 2022, David Bennett became the first living person to receive a genetically modified pig heart, surviving for two months before succumbing to complications that included a porcine virus. Since then, the field has learned from each case. Pig kidneys have been transplanted into brain-dead patients to study function and rejection, and in 2025 and 2026, living patients have received pig kidneys with progressively longer survival times. The 271-day survival reported in May 2026 represents a dramatic improvement and suggests that the immunological barriers, while not yet fully overcome, are becoming manageable.
Dr. Cooper, in his Frontiers interview, identified several remaining challenges. Swine leukocyte antigen (SLA) incompatibility — the pig equivalent of human HLA mismatching — remains poorly understood and may contribute to chronic rejection. Coagulation dysregulation, in which pig proteins interact abnormally with human clotting factors, can cause dangerous blood clots or bleeding. And the immunosuppressive regimens required to prevent rejection remain heavy, carrying risks of infection and malignancy that must be balanced against the benefits of transplantation.
Despite these challenges, the trajectory is unmistakable. The FDA is actively developing regulatory pathways for xenotransplantation clinical trials. Multiple companies have invested hundreds of millions of dollars in pig breeding facilities, genetic engineering platforms, and clinical infrastructure. And the scientific community has moved from asking whether xenotransplantation can work to asking how quickly it can be scaled.
For the families of the seventeen Americans who die every day waiting for an organ, the answer cannot come soon enough. Xenotransplantation may not yet be routine, but in 2026, it is no longer a dream. It is a clinical reality taking shape in operating rooms, research laboratories, and pig farms across the United States and beyond.
References: Cooper DKC. Xenotransplantation: lessons from history, genetic engineering, and early clinical experience. Frontiers in Transplantation. 8 May 2026. USA Today. “Doomed to die, one man chose a risky experiment that changed history.” 3 May 2026. Yahoo Finance/Post-Bulletin. eGenesis facility acquisition. 28 April 2026. National Kidney Foundation. Xenotransplantation update. May 2026.